When it comes to chemistry, understanding solubility rules is crucial for predicting how different compounds will behave in various solutions. A solubility rules chart can be a powerful tool for chemists, providing a quick and easy way to determine the solubility of a particular compound. As someone who's worked with these charts, I can attest to their usefulness. Here are some key points to keep in mind when using a solubility rules chart:
1. Nitrates Are Always Soluble
Nitrates, regardless of the metal ion they're paired with, are always soluble in water. This is a fundamental rule to remember, as it applies broadly across many different compounds. Whether you're working with sodium nitrate or calcium nitrate, you can be confident that it will dissolve in water.
2. Chlorides Are Generally Soluble, Except for Silver and Lead
Chlorides are soluble in most cases, but there are a couple of notable exceptions. Silver chloride and lead chloride are both insoluble, so it's essential to remember these exceptions when working with chlorides. This rule can help you predict the behavior of chlorides in different situations.
3. Sulfates Are Soluble, Except for Calcium, Strontium, and Barium
Sulfates are generally soluble, but there are a few metal ions that form insoluble sulfates. Calcium, strontium, and barium sulfates are all insoluble, so it's crucial to keep these exceptions in mind. This rule can help you anticipate how sulfates will behave in different solutions.
4. Carbonates Are Insoluble, Except for Alkali Metals and Ammonium
Carbonates are generally insoluble, but there are some exceptions. Alkali metals like sodium and potassium, as well as ammonium, form soluble carbonates. This rule can help you predict the behavior of carbonates in different situations and avoid unwanted precipitation.
5. Phosphates Are Insoluble, Except for Alkali Metals and Ammonium
Phosphates follow a similar pattern to carbonates, with most being insoluble except for those paired with alkali metals or ammonium. This rule is critical for working with phosphate compounds and anticipating their solubility in different solutions.
6. Hydroxides Are Insoluble, Except for Alkali Metals and Alkaline Earth Metals
Hydroxides are generally insoluble, but there are some exceptions. Alkali metals and alkaline earth metals form soluble hydroxides, which is an important distinction to remember. This rule can help you predict the behavior of hydroxides in different situations.
7. Oxides Are Insoluble, Except for Alkali Metals and Alkaline Earth Metals
Oxides follow a similar pattern to hydroxides, with most being insoluble except for those paired with alkali metals or alkaline earth metals. This rule is essential for working with oxide compounds and anticipating their solubility in different solutions.
8. Solubility Rules Can Be Affected by pH and Concentration
While solubility rules provide a general guideline, they can be influenced by factors like pH and concentration. In some cases, a compound may be more or less soluble depending on the specific conditions. It's essential to consider these factors when applying solubility rules in real-world situations.
9. Solubility Rules Are Not Always Absolute
It's essential to remember that solubility rules are not always absolute. There can be exceptions and variations depending on the specific compound and conditions. By recognizing these limitations, you can use solubility rules as a guideline while also considering other factors that may influence solubility.
10. Practice and Experience Are Key to Mastering Solubility Rules
Finally, mastering solubility rules takes practice and experience. The more you work with these charts and apply the rules in different situations, the more comfortable you'll become with predicting solubility. With time and practice, you'll develop a deeper understanding of how these rules work and how to apply them effectively.
If you are looking for Solubility Rules Chart in Word and Pdf formats - page 2 of 2 you've visit to the right place. We have 10 Images about Solubility Rules Chart in Word and Pdf formats - page 2 of 2 like Solubility Rules Chart - download free documents for PDF, Word and Excel, Ion Sheet with Solubility Rules-2 | PDF | Sulfate | Ammonium and also Solubility Rules Chart in Word and Pdf formats - page 2 of 2. Here you go:
Solubility Rules Chart In Word And Pdf Formats - Page 2 Of 2
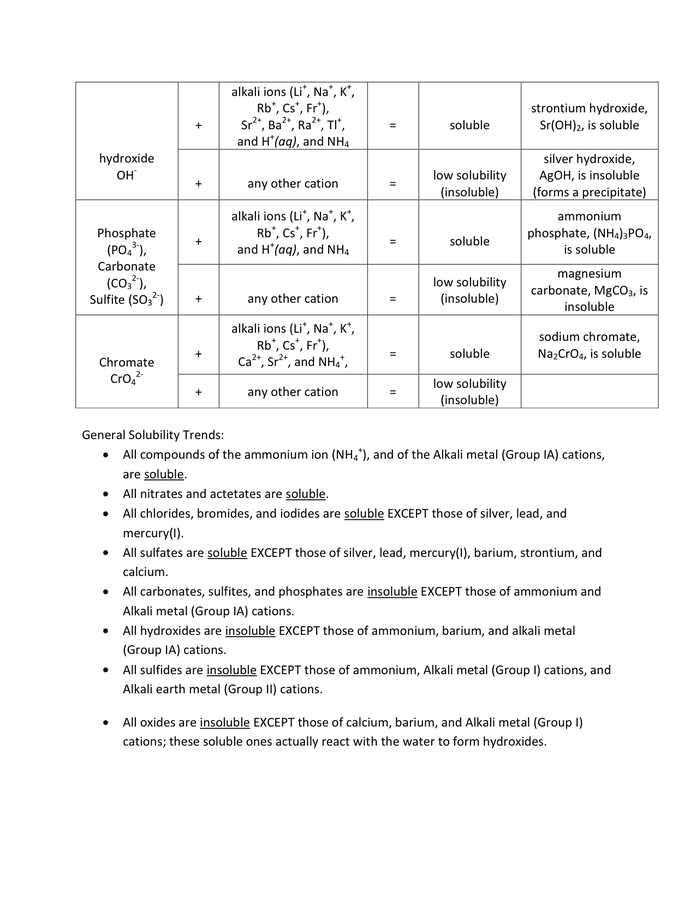 www.dexform.com
www.dexform.com
Solubility Rules Chart in Word and Pdf formats - page 2 of 2
Solubility Rules Chart - Download Chemistry Chart For Free PDF Or Word
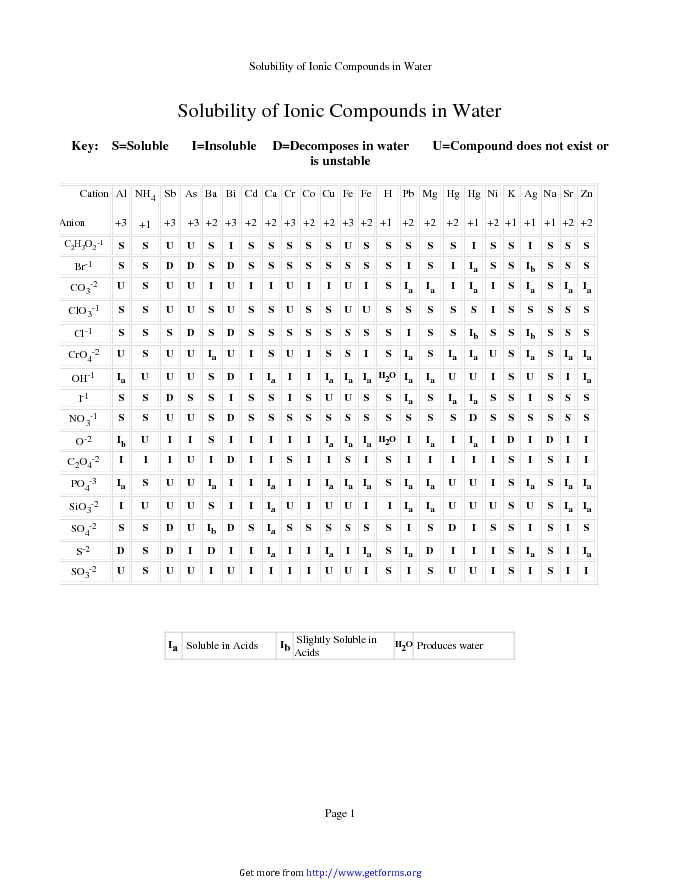 www.getforms.org
www.getforms.org
Solubility Rules Chart - download Chemistry Chart for free PDF or Word
Solubility Rules Chart - Download Free Documents For PDF, Word And Excel
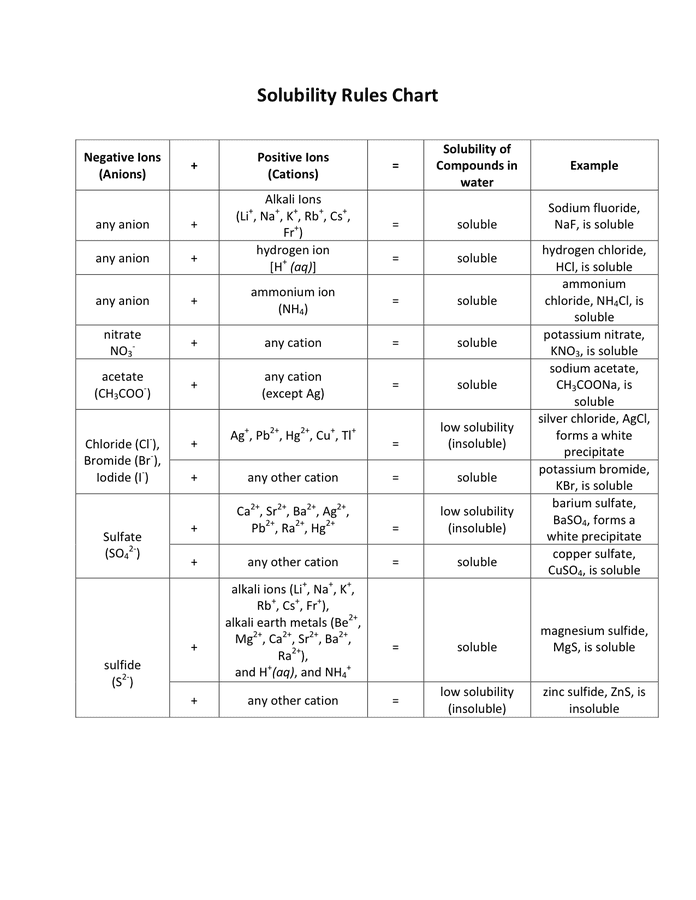 www.dexform.com
www.dexform.com
Solubility Rules Chart - download free documents for PDF, Word and Excel
Solubility Rules Diagram | Quizlet
 quizlet.com
quizlet.com
Solubility Rules Diagram | Quizlet
Solubility Rules Chart | Flinn Scientific
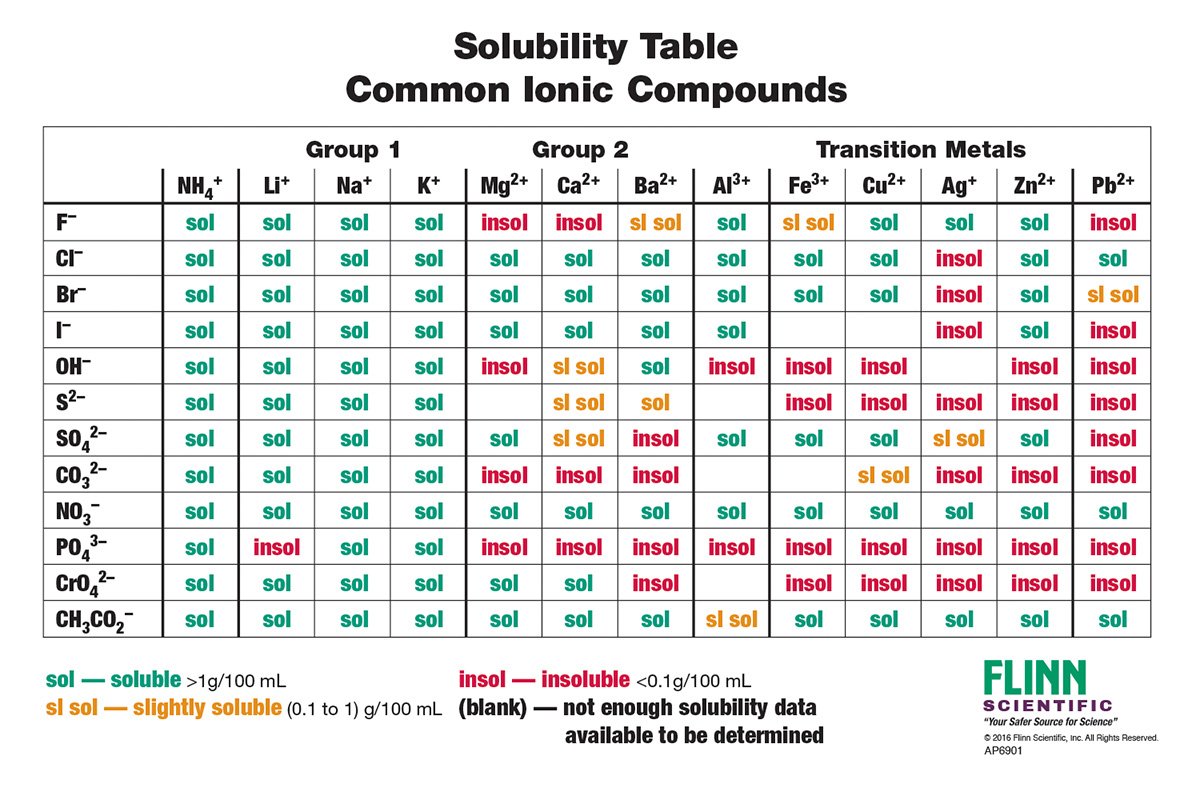 www.flinnsci.com
www.flinnsci.com
Solubility Rules Chart | Flinn Scientific
Solubility Rules Chart | Flinn Scientific
 www.flinnsci.com
www.flinnsci.com
Solubility Rules Chart | Flinn Scientific
Solubility Rules Chart
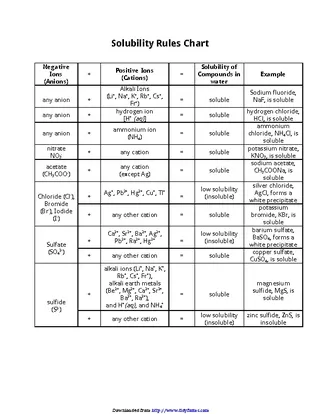 pdfsimpli.com
pdfsimpli.com
Solubility Rules Chart
Solubility Rules Chart | Flinn Scientific
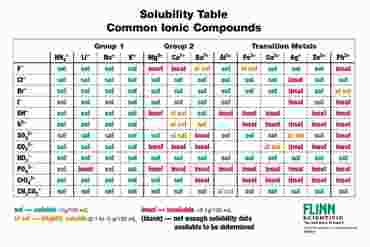 www.flinnsci.com
www.flinnsci.com
Solubility Rules Chart | Flinn Scientific
Ion Sheet With Solubility Rules-2 | PDF | Sulfate | Ammonium
 www.scribd.com
www.scribd.com
Ion Sheet with Solubility Rules-2 | PDF | Sulfate | Ammonium
Solubility Rules Chart - Download Chemistry Chart For Free PDF Or Word
 www.getforms.org
www.getforms.org
Solubility Rules Chart - download Chemistry Chart for free PDF or Word
Ion sheet with solubility rules-2. Solubility rules chart. solubility rules chart
