Polar and nonpolar charts are essential tools used in various fields, including chemistry, biology, and physics, to classify and understand the properties of molecules. These charts help scientists and researchers determine the polarity of a molecule, which is crucial in predicting its behavior, interaction with other molecules, and potential applications. In this article, we will explore the key aspects of polar and nonpolar charts, their importance, and how they are used in different fields.
1. Definition of Polar and Nonpolar Molecules
A polar molecule is a molecule that has a net dipole moment, meaning it has a slightly positive charge on one side and a slightly negative charge on the other. This occurs when there is an unequal sharing of electrons between atoms in a covalent bond. On the other hand, a nonpolar molecule has no net dipole moment, and the electrons are shared equally between atoms. Understanding the difference between polar and nonpolar molecules is crucial in predicting their properties and behavior.
2. Importance of Polar and Nonpolar Charts in Chemistry
Polar and nonpolar charts are essential in chemistry as they help scientists predict the properties of molecules, such as their boiling and melting points, solubility, and reactivity. By determining the polarity of a molecule, chemists can predict how it will interact with other molecules, which is crucial in understanding chemical reactions and developing new materials and products.
3. Types of Bonds that Determine Polarity
The type of bond between atoms in a molecule determines its polarity. Polar bonds are typically found in molecules with a difference in electronegativity between atoms, such as oxygen and hydrogen in a water molecule. Nonpolar bonds, on the other hand, are found in molecules with equal electronegativity between atoms, such as carbon and hydrogen in a methane molecule. Understanding the types of bonds that determine polarity is essential in predicting the properties of molecules.
4. Shape of Molecules and Polarity
The shape of a molecule also plays a crucial role in determining its polarity. Molecules with symmetrical shapes, such as carbon dioxide, are nonpolar, while molecules with asymmetrical shapes, such as water, are polar. The shape of a molecule can affect the distribution of electrons, leading to a net dipole moment and polarity.
5. Effects of Polarity on Intermolecular Forces
Polarity has a significant impact on intermolecular forces, which are the attractive and repulsive forces between molecules. Polar molecules experience stronger intermolecular forces, such as hydrogen bonding and dipole-dipole interactions, which affect their physical properties, such as boiling and melting points. Nonpolar molecules, on the other hand, experience weaker intermolecular forces, such as London dispersion forces.
6. Applications of Polar and Nonpolar Charts in Biology
Polar and nonpolar charts are used in biology to understand the properties of biomolecules, such as proteins and membranes. The polarity of these molecules affects their function and interaction with other molecules, which is crucial in understanding biological processes, such as cell signaling and transport.
7. Polarity and Solubility
Polarity affects the solubility of molecules in different solvents. Polar molecules are soluble in polar solvents, such as water, while nonpolar molecules are soluble in nonpolar solvents, such as hexane. Understanding the polarity of molecules is essential in predicting their solubility and developing new products and materials.
8. Determining Polarity using Chemical Formulas
Chemical formulas can be used to determine the polarity of a molecule. By analyzing the bonds between atoms and the shape of the molecule, scientists can predict its polarity. This information can be used to understand the properties and behavior of the molecule, as well as its potential applications.
9. Limitations of Polar and Nonpolar Charts
While polar and nonpolar charts are essential tools in understanding the properties of molecules, they have limitations. They do not provide information about the magnitude of polarity or the distribution of electrons in a molecule. Additionally, they do not account for the effects of temperature and pressure on the properties of molecules.
10. Conclusion and Future Directions
In conclusion, polar and nonpolar charts are essential tools in understanding the properties and behavior of molecules. By determining the polarity of a molecule, scientists can predict its properties, such as solubility, reactivity, and intermolecular forces. Future research directions include developing more accurate and comprehensive models of polarity and exploring the applications of polar and nonpolar charts in emerging fields, such as nanotechnology and biotechnology.
If you are searching about Polar vs Non-Polar Molecules Explained | PDF | Chemical Polarity you've came to the right page. We have 10 Images about Polar vs Non-Polar Molecules Explained | PDF | Chemical Polarity like Differentiating Polar and Non-Polar Molecules | PDF | Chemical Polarity, Polarity, Solutions, and Environmental Chemistry and also Polar vs Non-Polar Molecules Explained | PDF | Chemical Polarity. Read more:
Polar Vs Non-Polar Molecules Explained | PDF | Chemical Polarity
 www.scribd.com
www.scribd.com
Polar vs Non-Polar Molecules Explained | PDF | Chemical Polarity ...
Polar Vs Non-Polar Molecules Explained | PDF | Chemical Polarity
 www.scribd.com
www.scribd.com
Polar vs Non-Polar Molecules Explained | PDF | Chemical Polarity ...
Properties Of Polar And Nonpolar Molecules | PDF
 www.scribd.com
www.scribd.com
Properties of Polar and Nonpolar Molecules | PDF
Polar Vs NonPolar Complete Detailed Table | PDF | Chemical Polarity
 www.scribd.com
www.scribd.com
Polar Vs NonPolar Complete Detailed Table | PDF | Chemical Polarity ...
Polarity, Solutions, And Environmental Chemistry
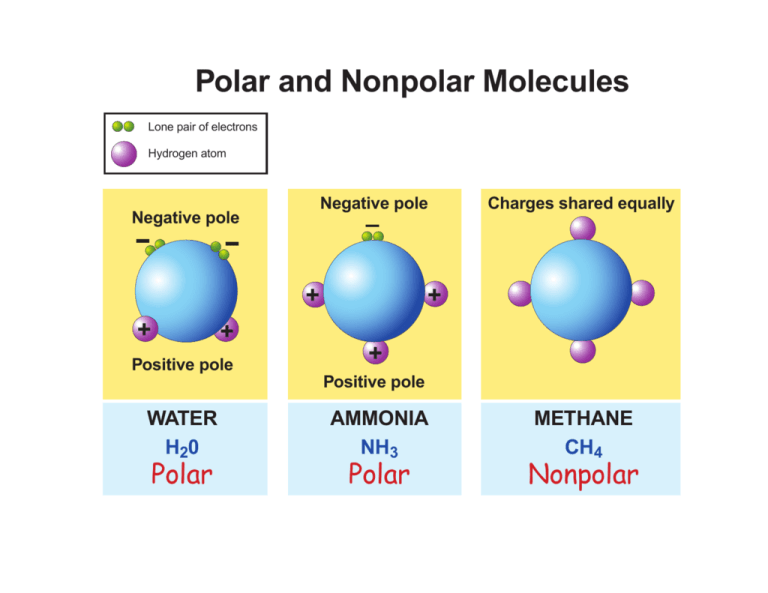 studylib.net
studylib.net
Polarity, Solutions, and Environmental Chemistry
Polar Vs. Non-Polar Bonds & Molecules | ChemTalk
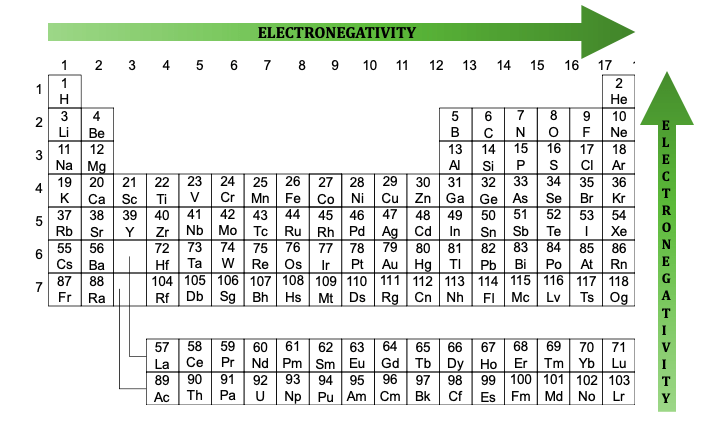 chemistrytalk.org
chemistrytalk.org
Polar vs. Non-Polar Bonds & Molecules | ChemTalk
Differentiating Polar And Non-Polar Molecules | PDF | Chemical Polarity
 www.scribd.com
www.scribd.com
Differentiating Polar and Non-Polar Molecules | PDF | Chemical Polarity ...
Difference Between Polar And Nonpolar Molecules | Definition, Formation
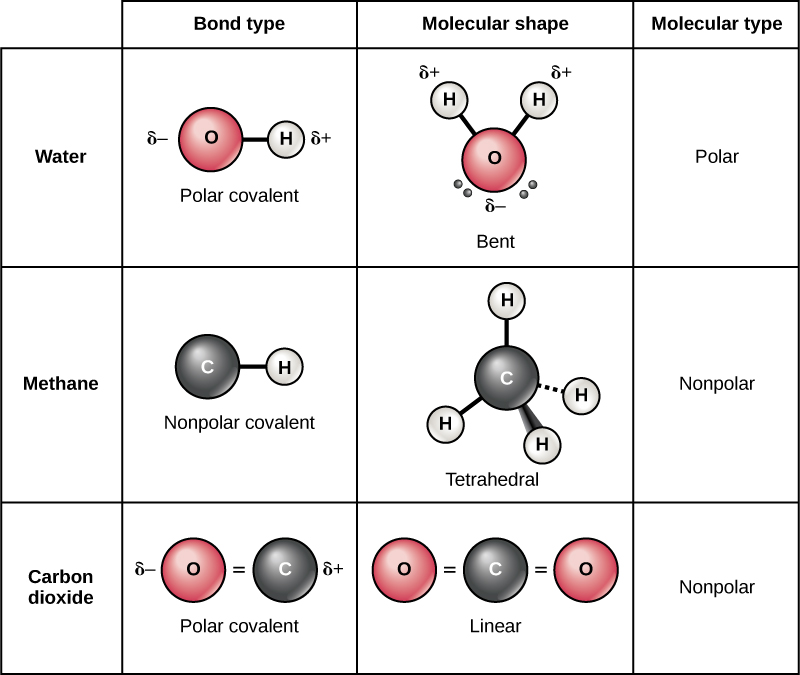 pediaa.com
pediaa.com
Difference Between Polar and Nonpolar Molecules | Definition, Formation ...
Polar Vs Non-Polar Molecules Explained | PDF | Chemical Polarity
 www.scribd.com
www.scribd.com
Polar vs Non-Polar Molecules Explained | PDF | Chemical Polarity ...
PPT - Polar Or Nonpolar PowerPoint Presentation - ID:3483667
 www.slideserve.com
www.slideserve.com
PPT - Polar or Nonpolar PowerPoint Presentation - ID:3483667
polar vs non-polar molecules explained. polar vs. non-polar bonds & molecules. Polarity, solutions, and environmental chemistry
