When dealing with the physical properties of substances, understanding the different stages they go through as they change from one state to another is fundamental. A phase change chart helps illustrate these transitions, providing crucial insights into the behavior of materials under varying conditions. Here's a breakdown of key points related to phase change charts, helping to clarify their importance and application in science.
1. Introduction to Phase Change
A phase change occurs when a substance moves from one state of matter to another - solid, liquid, or gas. This can happen due to changes in temperature or pressure. Phase change charts graphically represent these transitions, making it easier to visualize and understand the processes involved.
2. Solid-Liquid Phase Change (Melting)
Melting is the process where a solid turns into a liquid. This occurs when the temperature of the substance is increased to its melting point. The phase change chart will show this transition as a line separating the solid and liquid phases, with the exact melting point depending on the specific substance in question.
3. Liquid-Gas Phase Change (Vaporization)
Vaporization is the transition from a liquid to a gas. This can happen through evaporation (at the surface) or boiling (throughout the liquid). The chart will indicate the boiling point of a substance, which is the temperature at which vaporization occurs at standard atmospheric pressure.
4. Solid-Gas Phase Change (Sublimation)
Sublimation is a phase change where a solid directly transforms into a gas without going through the liquid phase. This process is less common and occurs in specific substances like iodine or dry ice (frozen carbon dioxide). Phase change charts will show this transition as a line connecting the solid and gas phases, skipping the liquid phase.
5. Gas-Liquid Phase Change (Condensation)
Condensation is the opposite of vaporization, where a gas changes back into a liquid. This process occurs when the gas cools down to its dew point, the temperature at which condensation happens. The phase change chart illustrates this transition as the gas state changing back to the liquid state upon cooling.
6. Gas-Solid Phase Change (Deposition)
Deposition is a phase change where a gas directly transforms into a solid without going through the liquid phase. This is the reverse process of sublimation. Water vapor directly turning into ice without becoming liquid water is an example of deposition. This transition is also represented on phase change charts, highlighting the direct transition from gas to solid.
7. Triple Point
The triple point is a unique condition on the phase change chart where all three phases (solid, liquid, and gas) of a substance coexist in equilibrium. This occurs at a specific temperature and pressure, which is different for each substance. The triple point is crucial because it defines the parameters under which phase changes can occur.
8. Critical Point
The critical point is another significant marker on the phase change chart, representing the temperature and pressure above which the distinction between the liquid and gas phases disappears. Beyond the critical point, the substance is considered a supercritical fluid, exhibiting properties of both liquids and gases.
9. Applications of Phase Change Charts
Understanding and using phase change charts has numerous practical applications in fields such as chemistry, physics, and engineering. They help in designing systems for refrigeration, power generation, and material processing, where the manipulation of phase changes is critical for efficiency and safety.
10. Conclusion
In conclusion, phase change charts are invaluable tools for visualizing and comprehending the transitions between different states of matter. By understanding these charts and the processes they illustrate, scientists and engineers can better design, predict, and control the behavior of substances under various conditions, leading to advancements in technology and our understanding of the physical world.
If you are looking for Phase Change Diagram - Labelled diagram you've visit to the right page. We have 10 Pictures about Phase Change Diagram - Labelled diagram like Phase Change - Role of Temperature and Pressure | PDF, Phase Change Chart Diagram | Quizlet and also Phase Change Chart check for understanding by StrellaBella | TPT. Here you go:
Phase Change Diagram - Labelled Diagram
Phase Change Diagram - Labelled diagram
Phase Change Chart
 fity.club
fity.club
Phase Change Chart
Phase Change Chart
 fity.club
fity.club
Phase Change Chart
Phase Change Diagrams — Overview Examples Expii, 57% OFF
 brunofuga.adv.br
brunofuga.adv.br
Phase Change Diagrams — Overview Examples Expii, 57% OFF
Phase Change Diagram - Mrs. Roundy Science
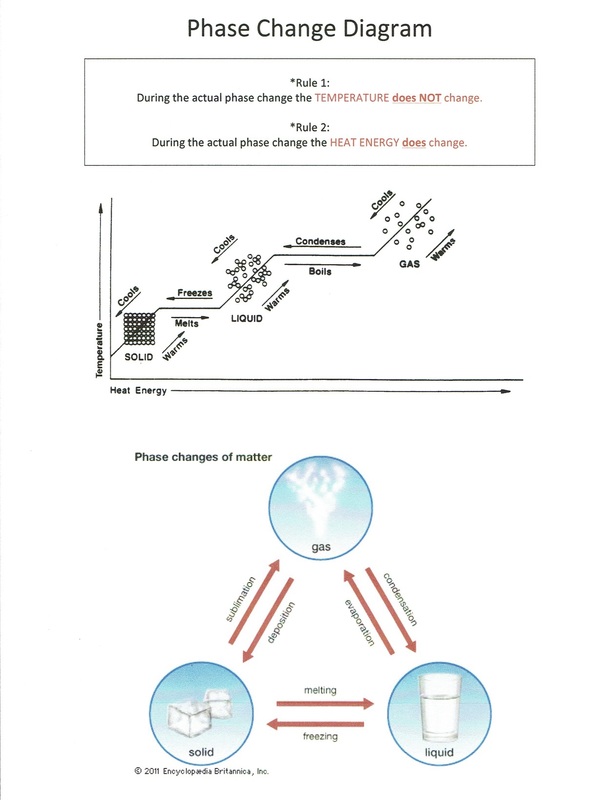 roundyscience.weebly.com
roundyscience.weebly.com
Phase Change Diagram - Mrs. Roundy Science
Phase Change - Role Of Temperature And Pressure | PDF
 www.scribd.com
www.scribd.com
Phase Change - Role of Temperature and Pressure | PDF
Phase Change Chart | Free Phase Change Chart Templates
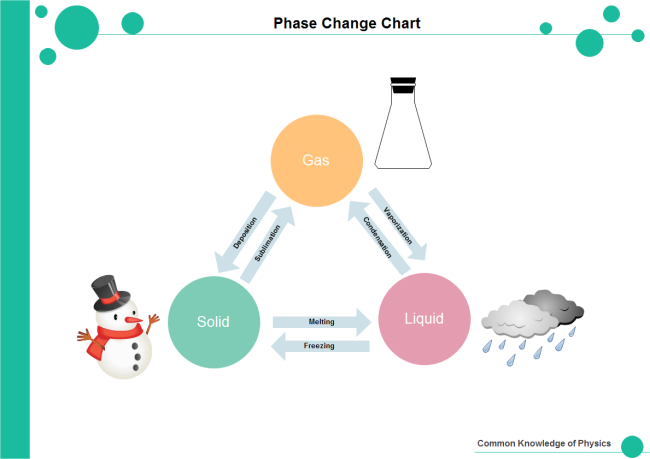 www.edrawsoft.com
www.edrawsoft.com
Phase Change Chart | Free Phase Change Chart Templates
Phase Change Chart Diagram | Quizlet
 quizlet.com
quizlet.com
Phase Change Chart Diagram | Quizlet
Phase Change Chart Check For Understanding By StrellaBella | TPT
 www.teacherspayteachers.com
www.teacherspayteachers.com
Phase Change Chart check for understanding by StrellaBella | TPT
Phase Change Chart
 fity.club
fity.club
Phase Change Chart
phase change chart check for understanding by strellabella. phase change diagrams — overview examples expii, 57% off. phase change chart
