When it comes to chemistry, one of the most important concepts to grasp is the idea of a limiting reactant. This is the reactant that will be completely used up in a chemical reaction, thereby limiting the amount of product that can be formed. Calculating the limiting reactant is crucial in determining the maximum amount of product that can be obtained from a given reaction. In this article, we will explore the steps involved in limiting reactant calculation and provide some tips to help you master this concept.
1. Understand the Concept of Limiting Reactant
The limiting reactant is the reactant that is present in the smallest stoichiometric amount, meaning that it will be completely consumed in the reaction. To identify the limiting reactant, you need to calculate the number of moles of each reactant and compare them to the coefficients in the balanced chemical equation. This will help you determine which reactant will be used up first.
2. Write Down the Balanced Chemical Equation
To calculate the limiting reactant, you need to start by writing down the balanced chemical equation for the reaction. This will give you the coefficients of each reactant and product, which are necessary for calculating the number of moles. Make sure to double-check that the equation is balanced, as this will affect your calculations.
3. Calculate the Number of Moles of Each Reactant
Once you have the balanced chemical equation, you can calculate the number of moles of each reactant. To do this, you will need to know the mass of each reactant in grams and the molar mass of each reactant. You can use the formula: moles = mass / molar mass. This will give you the number of moles of each reactant.
4. Compare the Number of Moles to the Coefficients
Now that you have the number of moles of each reactant, you can compare them to the coefficients in the balanced chemical equation. Divide the number of moles of each reactant by the coefficient of that reactant in the equation. This will give you a ratio of moles to coefficients, which will help you identify the limiting reactant.
5. Identify the Limiting Reactant
The reactant with the smallest ratio of moles to coefficients is the limiting reactant. This is the reactant that will be completely consumed in the reaction and will determine the maximum amount of product that can be formed. Make sure to identify the limiting reactant correctly, as this will affect your calculations.
6. Calculate the Maximum Amount of Product
Once you have identified the limiting reactant, you can calculate the maximum amount of product that can be formed. To do this, you will need to use the coefficient of the product in the balanced chemical equation and the number of moles of the limiting reactant. You can use the formula: moles of product = moles of limiting reactant x coefficient of product. This will give you the maximum amount of product that can be formed.
7. Consider the Percent Yield
In reality, the actual amount of product formed may be less than the theoretical maximum due to factors such as incomplete reactions and losses during purification. To account for this, you can calculate the percent yield, which is the ratio of the actual amount of product formed to the theoretical maximum. This will give you a more realistic estimate of the amount of product that can be formed.
8. Practice Limiting Reactant Problems
Calculating the limiting reactant can be challenging, so it's essential to practice problems to develop your skills. Start with simple problems and gradually move on to more complex ones. This will help you become more confident and proficient in calculating the limiting reactant.
9. Use Online Resources and Calculators
There are many online resources and calculators available that can help you with limiting reactant calculations. These can be useful tools to check your work and provide additional practice problems. However, make sure to understand the underlying concepts and not just rely on calculators to do the work for you.
10. Review and Apply the Concepts
Finally, it's essential to review and apply the concepts of limiting reactant calculation regularly. This will help you retain the information and develop a deeper understanding of the subject. Try to apply the concepts to real-world problems and scenarios, and seek help if you're struggling with any aspect of the calculation.
If you are looking for Limiting Reactant in the Stoichiometry of Chemical Reactions you've came to the right place. We have 10 Pictures about Limiting Reactant in the Stoichiometry of Chemical Reactions like Limiting Reactant Calculation | PDF, Limiting Reactant Explained: Step-by-Step Guide & Practice Problems and also Limiting Reactant Explained: Step-by-Step Guide & Practice Problems. Read more:
Limiting Reactant In The Stoichiometry Of Chemical Reactions
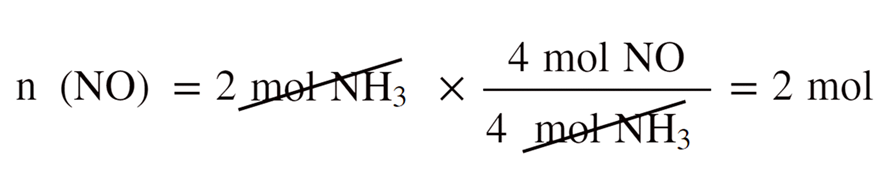 general.chemistrysteps.com
general.chemistrysteps.com
Limiting Reactant in the Stoichiometry of Chemical Reactions
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems
 www.chemdunn.com
www.chemdunn.com
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems ...
Limiting Reactant Stoichiometry: A Chemistry Worksheet | Made By
 worksheets.clipart-library.com
worksheets.clipart-library.com
Limiting Reactant Stoichiometry: A Chemistry Worksheet | Made By ...
Limiting Reactant Calculation | PDF
 www.scribd.com
www.scribd.com
Limiting Reactant Calculation | PDF
Limiting Reactant In The Stoichiometry Of Chemical Reactions
 general.chemistrysteps.com
general.chemistrysteps.com
Limiting Reactant in the Stoichiometry of Chemical Reactions
Limiting Reactant Calculator | Limiting Reagent And Theoretical Yield
 calculatorcorp.com
calculatorcorp.com
Limiting Reactant Calculator | Limiting Reagent And Theoretical Yield
Limiting Reactant | PDF
 www.scribd.com
www.scribd.com
Limiting Reactant | PDF
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems
 www.chemdunn.com
www.chemdunn.com
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems ...
Limiting Reactant - Solution Stoichiometry
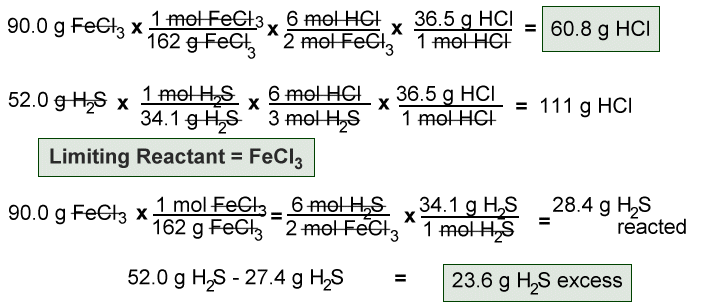 solutionstoichiometry.weebly.com
solutionstoichiometry.weebly.com
Limiting Reactant - Solution Stoichiometry
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems
 www.chemdunn.com
www.chemdunn.com
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems ...
Limiting reactant calculation. Limiting reactant explained: step-by-step guide & practice problems. limiting reactant in the stoichiometry of chemical reactions
