The ionization trend chart is a crucial tool in understanding the behavior of atoms and molecules, particularly in the context of chemistry and physics. It helps us visualize how the ionization energy of elements changes across different periods and groups in the periodic table. This chart is essential for students, researchers, and scientists alike, as it provides valuable insights into the atomic structure and chemical properties of various elements. In this article, we will delve into the key aspects of the ionization trend chart, exploring its significance and applications in the world of chemistry.
1. Introduction to Ionization Energy
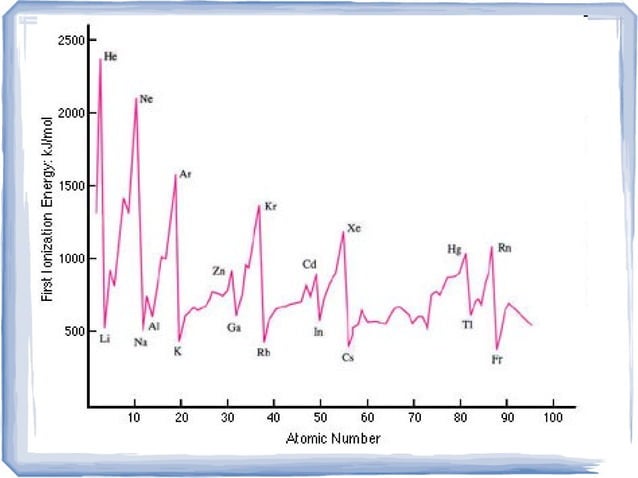
Ionization energy refers to the amount of energy required to remove an electron from an atom or molecule in its ground state. This energy is typically measured in electronvolts (eV) and is a critical parameter in understanding the chemical reactivity of elements. The ionization trend chart plots the ionization energy of elements against their atomic number, revealing patterns and trends that help us understand the underlying factors that influence this energy.
2. Periodic Trends in Ionization Energy
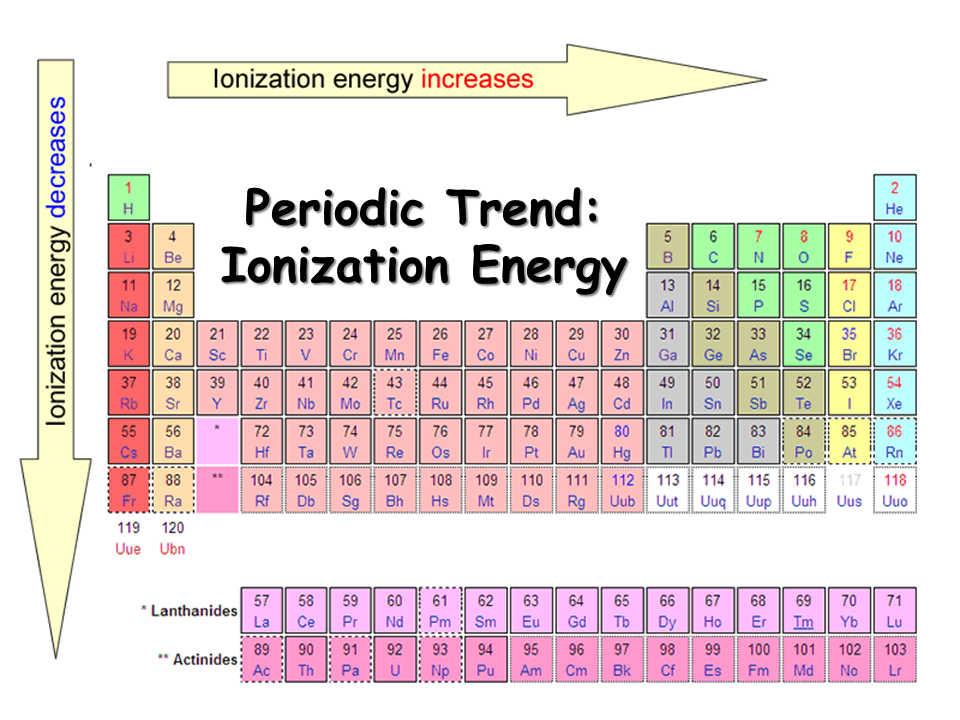
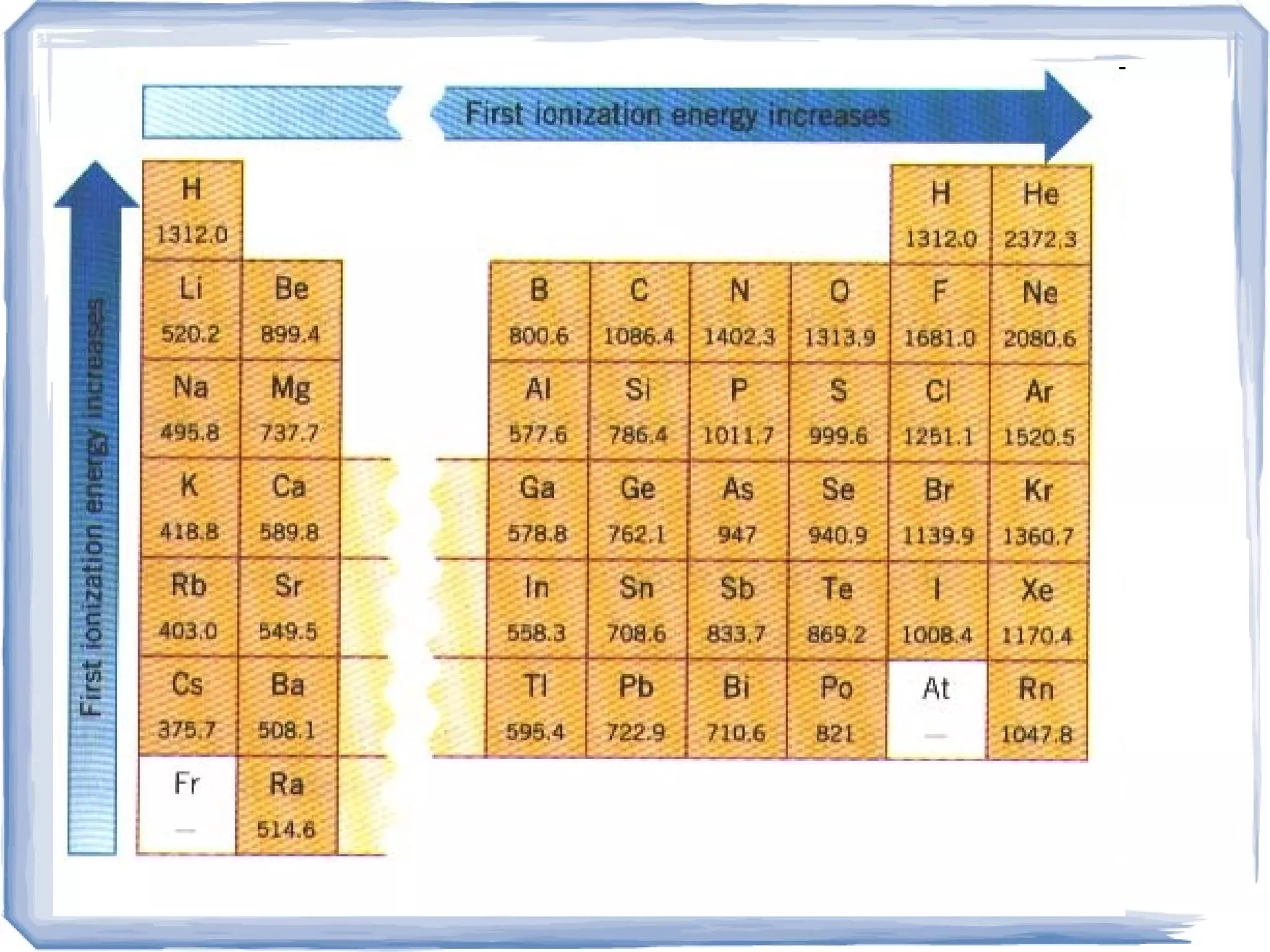
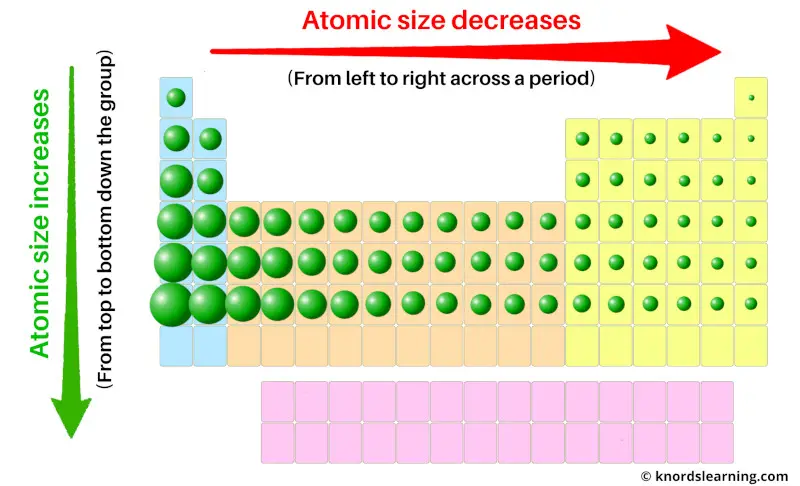
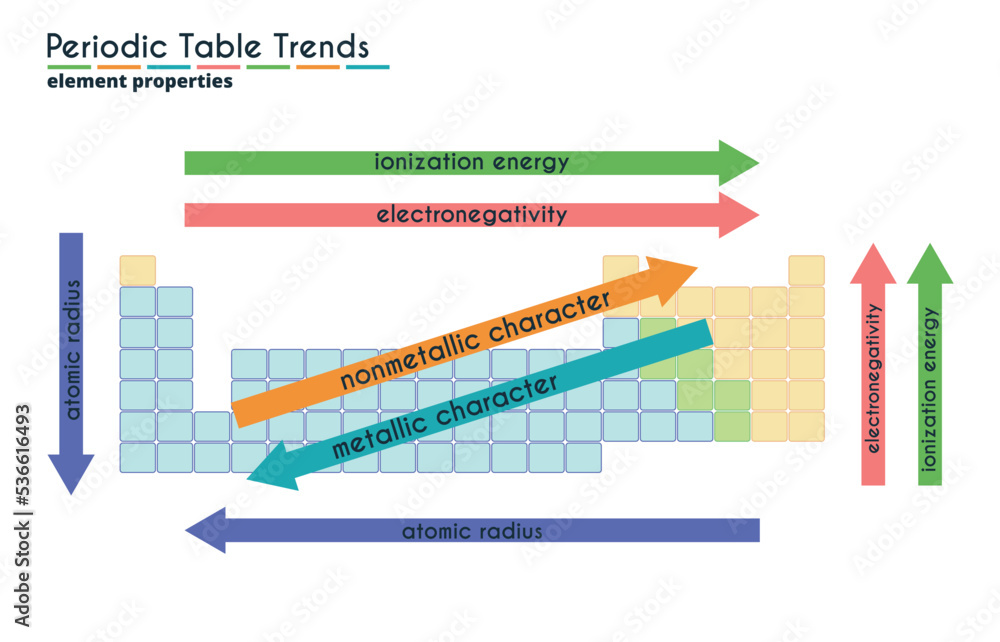
The ionization trend chart exhibits periodic trends, with ionization energy generally increasing from left to right across a period and decreasing down a group. This is due to the increasing effective nuclear charge and the shielding effect of inner electrons, which influence the ease with which an electron can be removed from an atom. These trends are essential in understanding the chemical properties of elements and their position in the periodic table.
3. Factors Influencing Ionization Energy
Several factors influence the ionization energy of an element, including the effective nuclear charge, electron configuration, and shielding effect. The effective nuclear charge refers to the net positive charge experienced by an electron in an atom, while the electron configuration determines the arrangement of electrons in an atom. The shielding effect, on the other hand, refers to the reduction in the effective nuclear charge due to the presence of inner electrons.
4. Group Trends in Ionization Energy
Ionization energy generally decreases down a group, as the atomic radius increases and the effective nuclear charge decreases. This is because the additional energy levels and electrons in the outermost energy level shield the inner electrons from the nucleus, making it easier to remove an electron. This trend is consistent across the periodic table, with some exceptions due to the presence of half-filled or completely filled subshells.
5. Period Trends in Ionization Energy
Ionization energy generally increases across a period, as the effective nuclear charge increases and the atomic radius decreases. This is because the additional protons in the nucleus increase the effective nuclear charge, making it more difficult to remove an electron. However, there are some exceptions to this trend, particularly in the transition metals and the noble gases.
6. Noble Gases and Ionization Energy
The noble gases, located in group 18 of the periodic table, have a unique electron configuration that makes them extremely stable. Their outermost energy level is completely filled, which results in a high ionization energy. This is because the removal of an electron from a noble gas atom requires a significant amount of energy to overcome the strong electrostatic attraction between the nucleus and the electrons.
7. Transition Metals and Ionization Energy
The transition metals, located in the d-block of the periodic table, exhibit a complex trend in ionization energy. Their ionization energy is generally lower than that of the noble gases, due to the presence of partially filled d-subshells. However, the ionization energy of transition metals can vary significantly depending on the specific element and its electron configuration.
8. Applications of Ionization Trend Chart
The ionization trend chart has numerous applications in chemistry and physics, including the prediction of chemical reactivity and the understanding of atomic structure. It is also essential in the study of thermodynamics, kinetics, and spectroscopy, as it provides valuable insights into the energy levels and electron configurations of atoms and molecules.
9. Limitations of Ionization Trend Chart
While the ionization trend chart is a powerful tool in understanding the behavior of atoms and molecules, it has some limitations. It does not account for the complexities of molecular systems or the effects of external factors such as temperature and pressure. Additionally, the chart is based on the assumption of a single electron being removed from an atom, which may not always be the case in real-world scenarios.
10. Conclusion and Future Directions
In conclusion, the ionization trend chart is a fundamental tool in understanding the behavior of atoms and molecules. Its applications are diverse, ranging from the prediction of chemical reactivity to the understanding of atomic structure. However, the chart also has some limitations, and future research should focus on developing more sophisticated models that can account for the complexities of molecular systems and external factors. By continuing to explore and refine the ionization trend chart, we can gain a deeper understanding of the underlying principles of chemistry and physics, leading to new discoveries and innovations in these fields.
If you are looking for Ionization Enthalpy - NEET Lab you've visit to the right place. We have 10 Images about Ionization Enthalpy - NEET Lab like Ionization Energy | Periodic Table Trends | ChemTalk, ionization energy trend | ODP and also ionization energy trend | ODP. Read more:
Ionization Enthalpy - NEET Lab
 neetlab.com
neetlab.com
Ionization Enthalpy - NEET Lab
Ionization Energy Trend | ODP
 www.slideshare.net
www.slideshare.net
ionization energy trend | ODP
Ionization Energy Trend | ODP
 www.slideshare.net
www.slideshare.net
ionization energy trend | ODP
Ionization Energy Trend | ODP
 www.slideshare.net
www.slideshare.net
ionization energy trend | ODP
Ionization Energy Trend In Periodic Table (Explained)
 knordslearning.com
knordslearning.com
Ionization Energy Trend in Periodic Table (Explained)
Ionization Energy Trend In Periodic Table (Explained)
 knordslearning.com
knordslearning.com
Ionization Energy Trend in Periodic Table (Explained)
Chart Of Each Elements Ionization Energy - Educational Chart Resources
 educationsurvey.steelcase.com
educationsurvey.steelcase.com
Chart Of Each Elements Ionization Energy - Educational Chart Resources
Ionization Energy | Periodic Table Trends | ChemTalk
Ionization Energy | Periodic Table Trends | ChemTalk
Ionization Energy Periodic Table Chart | Cabinets Matttroy
 cabinet.matttroy.net
cabinet.matttroy.net
Ionization Energy Periodic Table Chart | Cabinets Matttroy
What Is Ionization Energy? Definition And Trend
 sciencenotes.org
sciencenotes.org
What Is Ionization Energy? Definition and Trend
Ionization energy trend. Ionization energy. What is ionization energy? definition and trend
