Calculating yield percent is a crucial step in various fields, including chemistry, physics, and engineering. In Asian cultures, precision and attention to detail are highly valued, and yield percent calculation is no exception. To help you master this essential skill, we've put together a list of steps to follow. Whether you're a student, researcher, or industry professional, these tips will guide you through the process with ease.
1. Define Yield Percent and Its Importance
Yield percent, also known as percentage yield, is a measure of the amount of product obtained from a reaction or process compared to the theoretical maximum amount that could be obtained. It's a critical parameter in evaluating the efficiency and viability of a reaction or process. Understanding yield percent is essential in optimizing reaction conditions, reducing waste, and improving overall productivity.
2. Identify the Theoretical Yield
The theoretical yield is the maximum amount of product that can be obtained from a reaction based on the limiting reactant. To calculate the theoretical yield, you need to know the chemical equation, the stoichiometry of the reaction, and the amount of reactants used. The theoretical yield serves as a benchmark for evaluating the actual yield and calculating the yield percent.
3. Determine the Actual Yield
The actual yield is the amount of product obtained from a reaction or process. It's essential to measure the actual yield accurately, as it will be used to calculate the yield percent. The actual yield can be determined through various methods, including weighing, titration, or spectroscopy, depending on the nature of the product and the reaction.
4. Calculate the Yield Percent
Once you have the theoretical yield and the actual yield, you can calculate the yield percent using the formula: (actual yield / theoretical yield) x 100. This formula will give you a percentage value that represents the efficiency of the reaction or process.
5. Consider the Limiting Reactant
In a reaction, one reactant is often the limiting reactant, which determines the maximum amount of product that can be formed. When calculating yield percent, it's essential to identify the limiting reactant and use it to determine the theoretical yield. Failure to consider the limiting reactant can lead to incorrect yield percent calculations.
6. Account for Losses and Impurities
In real-world reactions and processes, losses and impurities can occur, affecting the actual yield and yield percent. It's crucial to account for these factors when calculating yield percent, as they can significantly impact the accuracy of the results. Common losses and impurities include side reactions, incomplete reactions, and contamination.
7. Use the Correct Units
When calculating yield percent, it's essential to use the correct units for the actual yield and theoretical yield. Ensure that both values are expressed in the same units, such as grams or moles, to avoid errors in the calculation.
8. Verify the Calculation
After calculating the yield percent, it's essential to verify the result to ensure accuracy. Check the calculation for errors, and if possible, compare the result with expected values or literature data. Verification is critical in ensuring the reliability of the yield percent calculation.
9. Interpret the Yield Percent Value
The yield percent value provides insight into the efficiency of a reaction or process. A high yield percent indicates a highly efficient process, while a low yield percent suggests areas for improvement. By interpreting the yield percent value, you can identify opportunities to optimize reaction conditions, reduce waste, and increase productivity.
10. Apply Yield Percent in Practice
Yield percent is a valuable tool in various fields, including chemistry, physics, and engineering. By applying yield percent calculations in practice, you can evaluate the efficiency of reactions and processes, optimize conditions, and improve overall productivity. Whether in research, industry, or academia, yield percent is an essential parameter in achieving success and driving innovation.
If you are looking for Percent Yield Formula | How to Calculate Yield - Lesson | Study.com you've visit to the right web. We have 10 Pics about Percent Yield Formula | How to Calculate Yield - Lesson | Study.com like The Best Way to Calculate Percent Yield in Chemistry - wikiHow, The Best Way to Calculate Percent Yield in Chemistry - wikiHow and also Percent Yield Formula | How to Calculate Yield - Lesson | Study.com. Here it is:
Percent Yield Formula | How To Calculate Yield - Lesson | Study.com
 worksheets.clipart-library.com
worksheets.clipart-library.com
Percent Yield Formula | How to Calculate Yield - Lesson | Study.com ...
How To Calculate Percent Yield « Math :: WonderHowTo
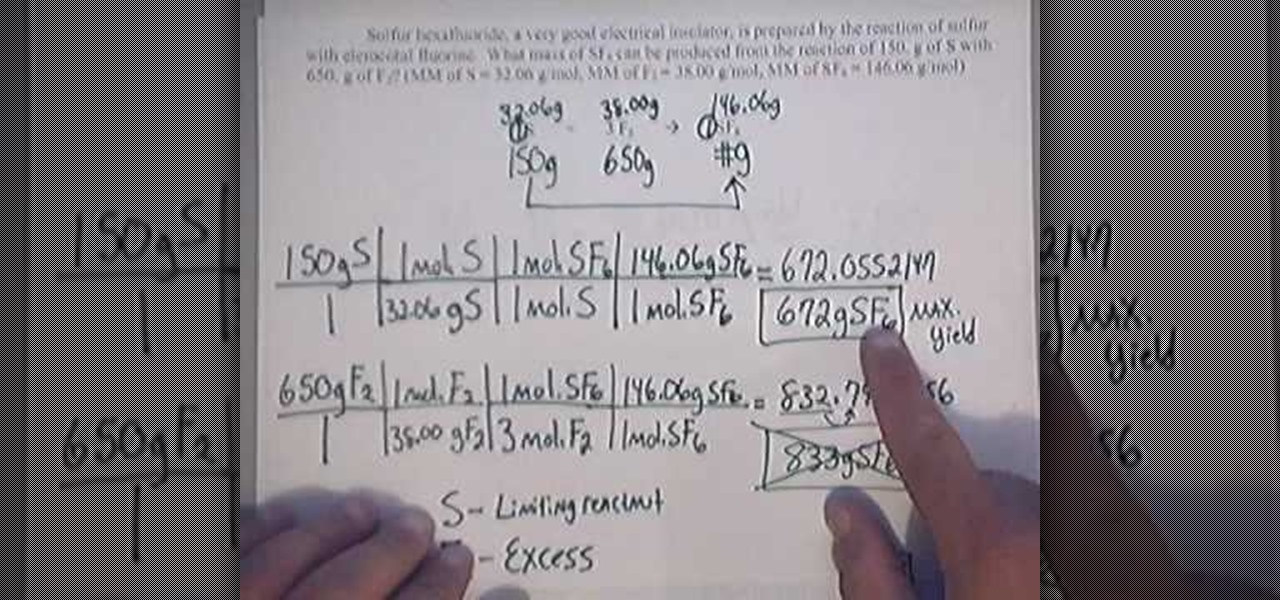 math.wonderhowto.com
math.wonderhowto.com
How to Calculate percent yield « Math :: WonderHowTo
Percent Yield Calculator - 100% Free Online Calculator
 calcgeek.com
calcgeek.com
Percent yield calculator - 100% Free online calculator
How Do You Calculate Percent Yield From Moles?
 scienceoxygen.com
scienceoxygen.com
How do you calculate percent yield from moles?
How To Calculate Percent Yield (Video)
 www.mometrix.com
www.mometrix.com
How to Calculate Percent Yield (Video)
The Best Way To Calculate Percent Yield In Chemistry - WikiHow
 www.wikihow.com
www.wikihow.com
The Best Way to Calculate Percent Yield in Chemistry - wikiHow
Percent Yield Calculator - Inch Calculator
 www.inchcalculator.com
www.inchcalculator.com
Percent Yield Calculator - Inch Calculator
How To Calculate Percent Yield In Chemistry: 13 Steps
 www.wikihow.com
www.wikihow.com
How to Calculate Percent Yield in Chemistry: 13 Steps
How To Calculate Percent Yield In Chemistry - Worksheets Library
How to Calculate Percent Yield in Chemistry - Worksheets Library
The Best Way To Calculate Percent Yield In Chemistry - WikiHow
 www.wikihow.com
www.wikihow.com
The Best Way to Calculate Percent Yield in Chemistry - wikiHow
percent yield calculator. The best way to calculate percent yield in chemistry. how to calculate percent yield (video)
