Calculating vapor pressure is a crucial concept in chemistry, particularly when dealing with liquids and gases. It's essential to understand the different methods and formulas used to determine vapor pressure to ensure accurate results. In this article, we'll delve into the various ways to calculate vapor pressure, providing you with a comprehensive guide to help you grasp this fundamental concept.
1. Understanding the Concept of Vapor Pressure
Vapor pressure is the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases at a given temperature. It's an important property of a substance that helps us understand its behavior and characteristics. To calculate vapor pressure, you need to understand the underlying principles and formulas that govern this concept.
2. Using the Clausius-Clapeyron Equation
The Clausius-Clapeyron equation is a fundamental formula used to calculate vapor pressure. The equation states that the vapor pressure of a substance is directly proportional to the exponential of the negative inverse of the temperature. Mathematically, it's expressed as: ln(P2/P1) = (ΔH/R) * (1/T1 - 1/T2), where P1 and P2 are the vapor pressures at temperatures T1 and T2, ΔH is the enthalpy of vaporization, and R is the gas constant.
3. Applying the Antoine Equation
The Antoine equation is another widely used formula to calculate vapor pressure. It's a semi-empirical equation that relates the vapor pressure of a substance to its temperature. The equation is expressed as: log10(P) = A - (B / (T + C)), where P is the vapor pressure, T is the temperature, and A, B, and C are substance-specific constants.
4. Utilizing the Vapor Pressure Chart
A vapor pressure chart is a graphical representation of the vapor pressure of a substance at different temperatures. It's a useful tool for quick reference and can be used to estimate the vapor pressure of a substance at a given temperature. However, it's essential to note that vapor pressure charts are substance-specific and may not be accurate for all temperatures and pressures.
5. Considering the Effects of Temperature
Temperature plays a significant role in determining the vapor pressure of a substance. As the temperature increases, the vapor pressure also increases. This is because higher temperatures provide more energy for the molecules to escape into the vapor phase, resulting in higher vapor pressure. Conversely, decreasing the temperature reduces the vapor pressure.
6. Accounting for Inter molecular Forces
Intermolecular forces, such as hydrogen bonding, dipole-dipole interactions, and London dispersion forces, can significantly impact the vapor pressure of a substance. Stronger intermolecular forces result in lower vapor pressure, as the molecules are more tightly bound together, making it more difficult for them to escape into the vapor phase.
7. Using the Raoult's Law
Raoult's law states that the vapor pressure of a solution is directly proportional to the mole fraction of the solvent. Mathematically, it's expressed as: P = (X * P0), where P is the vapor pressure of the solution, X is the mole fraction of the solvent, and P0 is the vapor pressure of the pure solvent.
8. Considering the Impact of Pressure
Pressure can also affect the vapor pressure of a substance. An increase in pressure can reduce the vapor pressure, as the molecules are compressed, making it more difficult for them to escape into the vapor phase. Conversely, a decrease in pressure can increase the vapor pressure, as the molecules have more space to move and escape into the vapor phase.
9. Using Online Calculators and Software
There are several online calculators and software available that can help you calculate vapor pressure. These tools can be useful for quick calculations and can provide accurate results. However, it's essential to understand the underlying principles and formulas to ensure that you're using the correct inputs and interpreting the results correctly.
10. Verifying Results with Experimental Data
Finally, it's essential to verify your calculated vapor pressure results with experimental data. This can be done by comparing your calculated values with published data or by conducting experiments to measure the vapor pressure of a substance. Verifying your results ensures that your calculations are accurate and reliable, providing you with confidence in your understanding of the concept of vapor pressure.
If you are looking for 3 Ways to Calculate Vapor Pressure - wikiHow you've visit to the right page. We have 10 Pics about 3 Ways to Calculate Vapor Pressure - wikiHow like To Calculate The Vapor Pressure Function | PDF, Chalk Talk: How To Calculate Vapor Pressure From Wet Bulb, 54% OFF and also How To Calculate The Vapor Pressure Of Water | Detroit Chinatown. Here you go:
3 Ways To Calculate Vapor Pressure - WikiHow
 www.wikihow.com
www.wikihow.com
3 Ways to Calculate Vapor Pressure - wikiHow
Chalk Talk: How To Calculate Vapor Pressure From Wet Bulb, 54% OFF
 www.pinnaxis.com
www.pinnaxis.com
Chalk Talk: How To Calculate Vapor Pressure From Wet Bulb, 54% OFF
Vapor Pressure: Definition, Formula, Examples, And Table
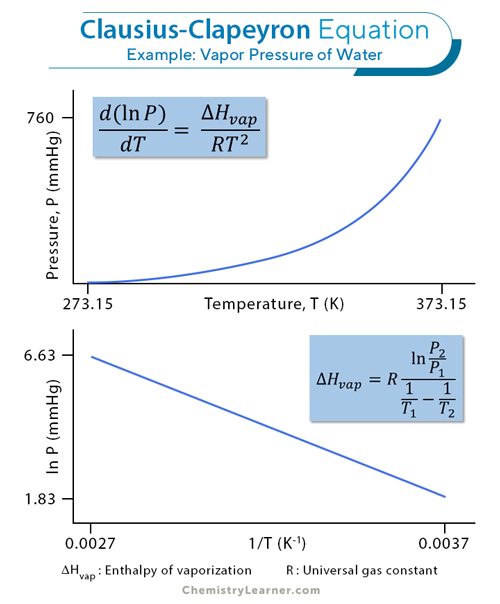 www.chemistrylearner.com
www.chemistrylearner.com
Vapor Pressure: Definition, Formula, Examples, and Table
Calculate Saturation Vapor Pressure
 sourcetable.com
sourcetable.com
Calculate Saturation Vapor Pressure
How To Calculate The Vapor Pressure Of Water | Detroit Chinatown
 detroitchinatown.org
detroitchinatown.org
How To Calculate The Vapor Pressure Of Water | Detroit Chinatown
Vapor Pressure Calculator (Clausius-Clapeyron Equation)
 molarity-calculator.org
molarity-calculator.org
Vapor Pressure Calculator (Clausius-Clapeyron Equation)
To Calculate The Vapor Pressure Function | PDF
 www.scribd.com
www.scribd.com
To Calculate The Vapor Pressure Function | PDF
Solved Calculate Vapor Pressure Solve The Clausius-Claperon | Chegg.com
 www.chegg.com
www.chegg.com
Solved Calculate vapor pressure Solve the Clausius-Claperon | Chegg.com
3 Ways To Calculate Vapor Pressure - WikiHow
 www.wikihow.com
www.wikihow.com
3 Ways to Calculate Vapor Pressure - wikiHow
Vapor Pressure: Definition, Formula, Examples, And Table
 www.chemistrylearner.com
www.chemistrylearner.com
Vapor Pressure: Definition, Formula, Examples, and Table
3 ways to calculate vapor pressure. calculate saturation vapor pressure. Vapor pressure calculator (clausius-clapeyron equation)
