Calculating valence electrons is a crucial concept in chemistry, as it helps determine the chemical properties and reactivity of an element. Valence electrons are the outermost electrons in an atom, and they play a significant role in forming chemical bonds. In this article, we will explore the steps to calculate valence electrons and provide a comprehensive guide for students and chemistry enthusiasts.
1. Determine the Atomic Number of the Element
The atomic number of an element is the number of protons present in the nucleus of an atom. To calculate valence electrons, you need to know the atomic number of the element. You can find the atomic number of an element by looking at the periodic table. The atomic number is usually represented by the symbol Z.
2. Find the Electron Configuration of the Element
The electron configuration of an element is the arrangement of electrons in an atom. You can find the electron configuration of an element by looking at the periodic table or using an online electron configuration calculator. The electron configuration is usually represented in a shorthand notation, such as 1s2 2s2 2p6.
3. Identify the Outermost Energy Level
The outermost energy level of an atom is the energy level that contains the valence electrons. The outermost energy level is usually the highest energy level that is occupied by electrons. You can identify the outermost energy level by looking at the electron configuration of the element.
4. Determine the Number of Valence Electrons in the Outermost Energy Level
Once you have identified the outermost energy level, you need to determine the number of valence electrons in that energy level. You can do this by counting the number of electrons in the outermost energy level. For example, if the electron configuration of an element is 1s2 2s2 2p6, the outermost energy level is the 2nd energy level, and it contains 8 electrons (2s2 2p6).
5. Consider the Noble Gas Core
The noble gas core is the inner energy levels of an atom that are completely filled with electrons. When calculating valence electrons, you can ignore the noble gas core, as it does not participate in chemical bonding. The noble gas core is usually represented in a shorthand notation, such as [Ne] or [Ar].
6. Account for D-Block and F-Block Elements
D-block and F-block elements have a different electron configuration than s-block and p-block elements. When calculating valence electrons for D-block and F-block elements, you need to consider the electrons in the d and f orbitals. These electrons can participate in chemical bonding and are considered valence electrons.
7. Calculate the Number of Valence Electrons for Ions
When an atom forms an ion, it gains or loses electrons. To calculate the number of valence electrons for an ion, you need to consider the number of electrons gained or lost. For example, if a sodium atom loses one electron to form a sodium ion, the number of valence electrons is zero.
8. Consider the Exceptions to the Octet Rule
The octet rule states that atoms tend to gain or lose electrons to form a full outer energy level with eight electrons. However, there are some exceptions to this rule, such as hydrogen, which can form a stable molecule with only two electrons. When calculating valence electrons, you need to consider these exceptions.
9. Use the Periodic Table to Verify the Calculation
The periodic table can be a useful tool for verifying the calculation of valence electrons. By looking at the periodic table, you can see the electron configuration of an element and the number of valence electrons. This can help you check your calculation and ensure that it is correct.
10. Practice with Different Elements
Calculating valence electrons can be a complex process, and it requires practice to become proficient. To improve your skills, try calculating the valence electrons for different elements, including s-block, p-block, d-block, and f-block elements. With practice, you will become more comfortable with the process and be able to calculate valence electrons with ease.
If you are looking for How To Determine Valence Electrons With Periodic Table - Infoupdate.org you've came to the right page. We have 10 Pics about How To Determine Valence Electrons With Periodic Table - Infoupdate.org like How Do You Calculate The Number of Valence Electrons in An Atom | PDF, Valence Electrons - Characteristics and Determination of Valence Electrons and also How To Determine Valence Electrons With Periodic Table - Infoupdate.org. Read more:
How To Determine Valence Electrons With Periodic Table - Infoupdate.org
 infoupdate.org
infoupdate.org
How To Determine Valence Electrons With Periodic Table - Infoupdate.org
How To Find Valence Electrons With The Periodic Table
 www.wikihow.com
www.wikihow.com
How to Find Valence Electrons with the Periodic Table
How To Find Valence Electrons With The Periodic Table
 www.wikihow.com
www.wikihow.com
How to Find Valence Electrons with the Periodic Table
How To Find Valence Electrons With The Periodic Table
 www.wikihow.com
www.wikihow.com
How to Find Valence Electrons with the Periodic Table
How Do You Calculate The Number Of Valence Electrons In An Atom | PDF
 www.scribd.com
www.scribd.com
How Do You Calculate The Number of Valence Electrons in An Atom | PDF ...
How To Determine Valence Electrons With Periodic Table - Infoupdate.org
 infoupdate.org
infoupdate.org
How To Determine Valence Electrons With Periodic Table - Infoupdate.org
Valence Electrons - Characteristics And Determination Of Valence Electrons
 byjus.com
byjus.com
Valence Electrons - Characteristics and Determination of Valence Electrons
How To Determine Valence Electrons With Periodic Table - Infoupdate.org
 infoupdate.org
infoupdate.org
How To Determine Valence Electrons With Periodic Table - Infoupdate.org
Periodic Table With Valence Electrons (Image) - Pediabay
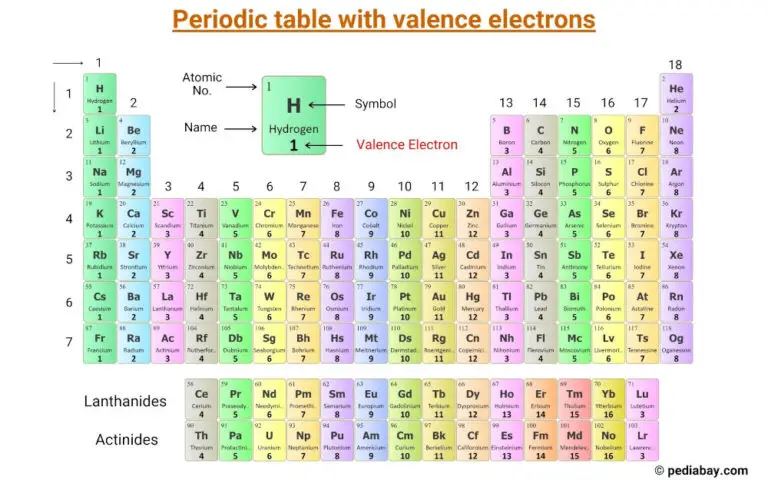 pediabay.com
pediabay.com
Periodic Table with Valence Electrons (Image) - Pediabay
Valence Electrons
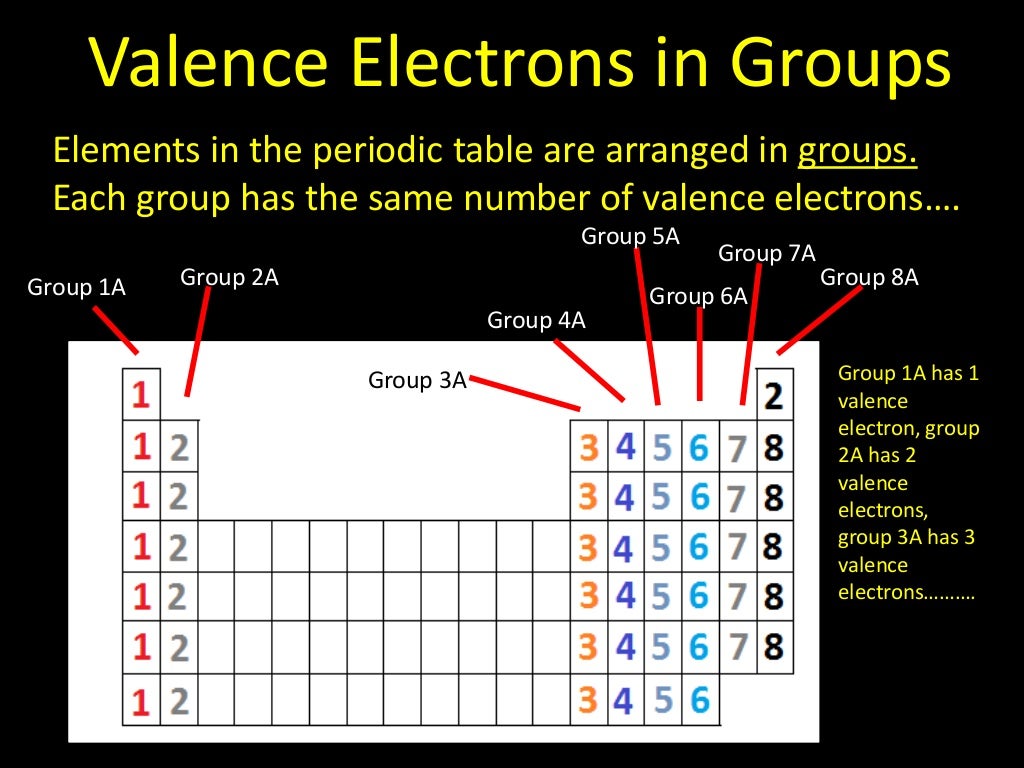 www.slideshare.net
www.slideshare.net
Valence Electrons
How to find valence electrons with the periodic table. Periodic table with valence electrons (image). How to determine valence electrons with periodic table
