Calculating the number of neutrons in an atom can be a daunting task, especially for those who are new to chemistry. However, with the right steps and a basic understanding of atomic structure, you can easily determine the number of neutrons in an atom. In this article, we will guide you through the process of calculating neutrons in a straightforward and easy-to-follow manner.
1. Determine the Atomic Number
The atomic number of an element is the number of protons present in the nucleus of an atom. To calculate the number of neutrons, you first need to know the atomic number of the element. You can find the atomic number on the periodic table, which is usually denoted by the symbol Z.
2. Determine the Mass Number
The mass number of an element is the total number of protons and neutrons present in the nucleus of an atom. The mass number is usually denoted by the symbol A. You can find the mass number on the periodic table or by using a mass spectrometer.
3. Use the Formula: Neutrons = Mass Number - Atomic Number
Now that you have the atomic number and the mass number, you can use the formula to calculate the number of neutrons. The formula is simple: Neutrons = Mass Number - Atomic Number. By subtracting the atomic number from the mass number, you can determine the number of neutrons in an atom.
4. Consider the Isotopes of an Element
Isotopes are atoms of the same element that have the same atomic number but different mass numbers. This means that isotopes have the same number of protons but different numbers of neutrons. When calculating the number of neutrons, you need to consider the specific isotope of the element you are working with.
5. Look for the Number of Neutrons in a Neutral Atom
A neutral atom has an equal number of protons and electrons. When calculating the number of neutrons, you should look for the number of neutrons in a neutral atom. This is because a neutral atom has no overall charge, and the number of protons and electrons is balanced.
6. Be Aware of the Atomic Mass Unit (amu)
The atomic mass unit (amu) is a unit of mass used to express the mass of an atom or molecule. The amu is defined as one-twelfth the mass of a carbon-12 atom. When calculating the number of neutrons, you may need to convert the atomic mass from amu to grams or kilograms.
7. Use Online Tools or Periodic Tables to Find the Number of Neutrons
There are many online tools and periodic tables available that can help you find the number of neutrons in an atom. These resources can save you time and effort, especially when working with complex calculations or rare isotopes.
8. Practice Calculating Neutrons with Sample Problems
Practicing with sample problems is an effective way to master the skill of calculating neutrons. You can find sample problems online or in chemistry textbooks. Start with simple problems and gradually move on to more complex ones to build your confidence and understanding.
9. Understand the Concept of Nuclear Stability
Nuclear stability refers to the balance between the number of protons and neutrons in an atom. When calculating the number of neutrons, it's essential to understand the concept of nuclear stability and how it affects the atom's structure and properties.
10. Review and Apply the Concepts to Real-World Scenarios
Finally, it's crucial to review and apply the concepts of calculating neutrons to real-world scenarios. This will help you understand the practical applications of chemistry and the importance of neutrons in various fields, such as nuclear energy, medicine, and materials science.
If you are looking for Calculating Protons, Electrons, and Neutrons | PDF you've came to the right page. We have 10 Pictures about Calculating Protons, Electrons, and Neutrons | PDF like Calculating Protons, Electrons, and Neutrons | PDF, Tutorial 22 Calculation of Number of Protons, Electrons and Neutrons and also Solved We can calculate the number of neutrons and protons | Chegg.com. Read more:
Calculating Protons, Electrons, And Neutrons | PDF
 www.scribd.com
www.scribd.com
Calculating Protons, Electrons, and Neutrons | PDF
[Solved] Calculate The Number Of Protons, Neutrons And Electrons In _{..
![[Solved] Calculate the number of protons, neutrons and electrons in _{..](https://classroom-notes.cdn.askfilo.com/thumb_classroom_29378925_3YQZB.jpeg) askfilo.com
askfilo.com
[Solved] Calculate the number of protons, neutrons and electrons in _{..
How Do You Calculate Neutrons? – ChemCafe — Science, Chemistry And
 chemcafe.net
chemcafe.net
How do you calculate neutrons? – ChemCafe — science, chemistry and ...
How Do You Calculate Neutrons - Oreate AI Blog
 www.oreateai.com
www.oreateai.com
How Do You Calculate Neutrons - Oreate AI Blog
How To Calculate The Number Of Protons, Neutrons And Electrons
 tnhelearning.edu.vn
tnhelearning.edu.vn
How to Calculate the Number of Protons, Neutrons and Electrons
Solved: The Formula Used To Calculate The Number Of Neutrons Is: Atomic
Solved: The formula used to calculate the number of neutrons is: Atomic ...
Solved We Can Calculate The Number Of Neutrons And Protons | Chegg.com
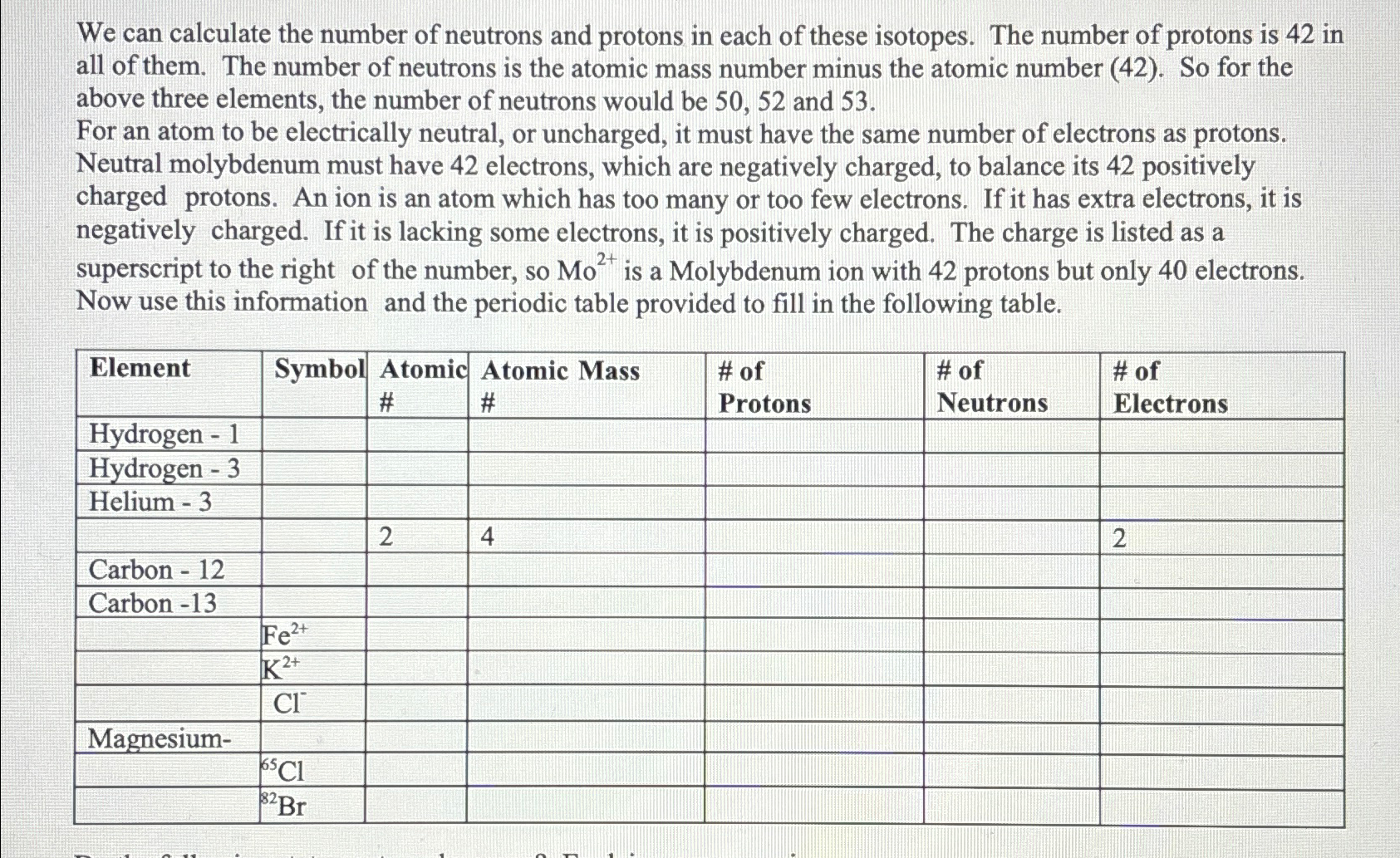 www.chegg.com
www.chegg.com
Solved We can calculate the number of neutrons and protons | Chegg.com
Solved: Calculating Protons, Neutrons And Electrons Calculate Protons
Solved: Calculating Protons, Neutrons and Electrons Calculate protons ...
Tutorial 22 Calculation Of Number Of Protons, Electrons And Neutrons
 www.scribd.com
www.scribd.com
Tutorial 22 Calculation of Number of Protons, Electrons and Neutrons ...
SECTION-A (CHEMISTRY)51. Calculate Number Of Neutrons Present In 12×1..
 askfilo.com
askfilo.com
SECTION-A (CHEMISTRY)51. Calculate number of neutrons present in 12×1..
Section-a (chemistry)51. calculate number of neutrons present in 12×1... Solved we can calculate the number of neutrons and protons. How to calculate the number of protons, neutrons and electrons
