Calculating solubility is a fundamental concept in chemistry that can be a bit tricky to grasp, but don't worry, we've got you covered. Solubility refers to the maximum amount of a substance that can dissolve in a given amount of solvent at a particular temperature. In this post, we'll break down the steps to calculate solubility in a way that's easy to understand and apply. So, let's dive in and explore the world of solubility calculations.
1. Understand the Solubility Concept
Before we start calculating solubility, it's essential to understand what it represents. Solubility is usually expressed in units of concentration, such as grams per liter (g/L) or moles per liter (mol/L). It's a measure of how much of a substance can dissolve in a solvent before the solution becomes saturated. Knowing this concept is crucial to making accurate calculations.
2. Identify the Solvent and Solute
To calculate solubility, you need to know the identity of the solvent and the solute. The solvent is the substance that does the dissolving, while the solute is the substance being dissolved. Common solvents include water, ethanol, and acetone, while solutes can be salts, sugars, or other compounds. Make sure you know which is which in your experiment or problem.
3. Determine the Temperature
Temperature plays a significant role in solubility calculations. The solubility of a substance can increase or decrease with temperature, depending on the specific compound. Make sure you know the temperature at which you're working, as this will affect your calculations. Some substances are more soluble at higher temperatures, while others are more soluble at lower temperatures.
4. Choose the Right Unit of Measurement
When calculating solubility, you need to choose the right unit of measurement. Common units include grams per liter (g/L), moles per liter (mol/L), or millimoles per liter (mmol/L). The unit you choose will depend on the specific problem or experiment you're working on. Make sure you're consistent in your units to avoid conversion errors.
5. Use Solubility Tables or Graphs
Solubility tables or graphs can be a great resource when calculating solubility. These tables or graphs provide the solubility of a substance at different temperatures, usually in a specific solvent. By referencing these tables or graphs, you can quickly determine the solubility of a substance without having to do complex calculations.
6. Calculate Molar Solubility
Molar solubility refers to the number of moles of a substance that can dissolve in a liter of solvent. To calculate molar solubility, you need to know the molar mass of the substance and the solubility in grams per liter. You can use the formula: molar solubility = solubility (g/L) / molar mass.
7. Consider the Effects of Pressure
Pressure can also affect solubility, especially for gases. An increase in pressure can increase the solubility of a gas in a liquid, while a decrease in pressure can decrease solubility. However, for most solids and liquids, pressure has a negligible effect on solubility.
8. Account for Ionization
Some substances, such as salts, can ionize in solution, which affects their solubility. When a salt ionizes, it breaks into its component ions, which can increase its solubility. You need to account for ionization when calculating the solubility of these substances.
9. Use the Solubility Product Constant (Ksp)
The solubility product constant (Ksp) is a measure of the equilibrium between a solid substance and its ions in solution. You can use Ksp to calculate the solubility of a substance, especially for salts and other ionic compounds. The Ksp value is specific to each substance and can be found in reference tables or calculated experimentally.
10. Verify Your Calculations
Finally, it's essential to verify your calculations to ensure accuracy. Check your units, make sure you've accounted for all the factors that affect solubility, and double-check your math. By following these steps and verifying your calculations, you can confidently determine the solubility of a substance and take your chemistry skills to the next level.
If you are searching about Solved B. Given Ksp Calculate Solubility Example: What is | Chegg.com you've came to the right web. We have 10 Pictures about Solved B. Given Ksp Calculate Solubility Example: What is | Chegg.com like How To Calculate Molar Solubility, How Do You Calculate the Solubility - Oreate AI Blog and also how to calculate the molar solubility from Ksp values??? - WizEdu. Here you go:
Solved B. Given Ksp Calculate Solubility Example: What Is | Chegg.com
 www.chegg.com
www.chegg.com
Solved B. Given Ksp Calculate Solubility Example: What is | Chegg.com
How To Calculate Solubility G 100g - Design Talk
 design.udlvirtual.edu.pe
design.udlvirtual.edu.pe
How To Calculate Solubility G 100g - Design Talk
SOLVED: Steps Needed Question 1: Calculate Molar Solubility And
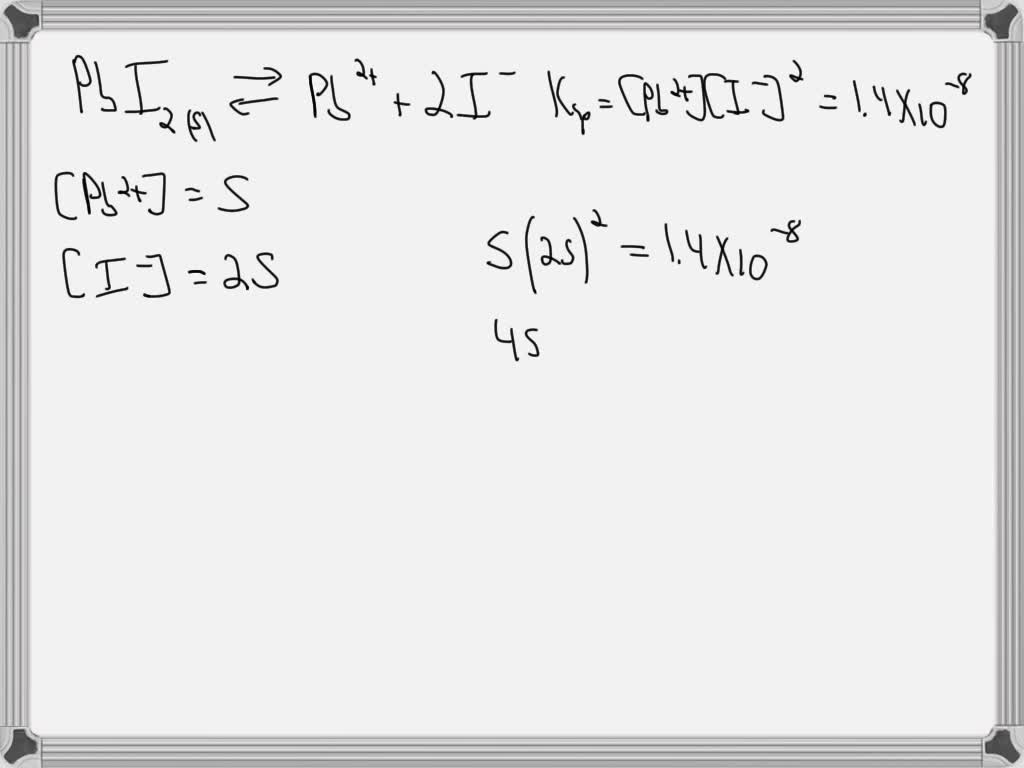 www.numerade.com
www.numerade.com
SOLVED: Steps needed Question 1: Calculate molar solubility and ...
How To Calculate Molar Solubility
 scising.com
scising.com
How To Calculate Molar Solubility
How Do You Calculate The Solubility - Oreate AI Blog
 www.oreateai.com
www.oreateai.com
How Do You Calculate the Solubility - Oreate AI Blog
Calculate The Solubility Of A Molecule Using Ksp Values Question If A
 www.homeworklib.com
www.homeworklib.com
Calculate the solubility of a molecule using Ksp values Question If a ...
Solved B. Given Ksp Calculate Solubility Example: What Is | Chegg.com
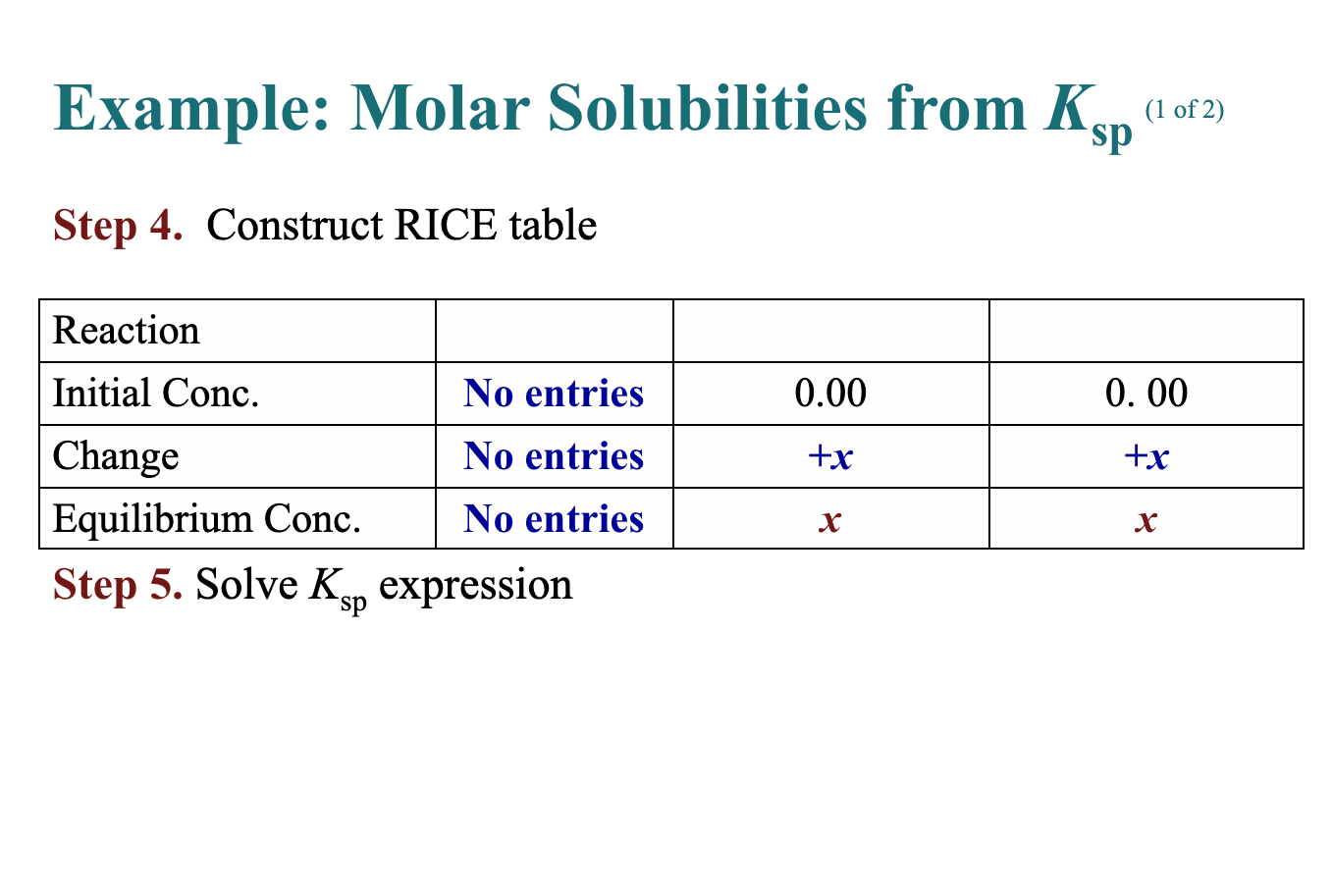 www.chegg.com
www.chegg.com
Solved B. Given Ksp Calculate Solubility Example: What is | Chegg.com
Calculate The Solubility Of A Molecule Using Ksp Values Question If A
 www.homeworklib.com
www.homeworklib.com
Calculate the solubility of a molecule using Ksp values Question If a ...
How To Calculate The Molar Solubility From Ksp Values??? - WizEdu
 wizedu.com
wizedu.com
how to calculate the molar solubility from Ksp values??? - WizEdu
How To Calculate Solubility | Chegg.com
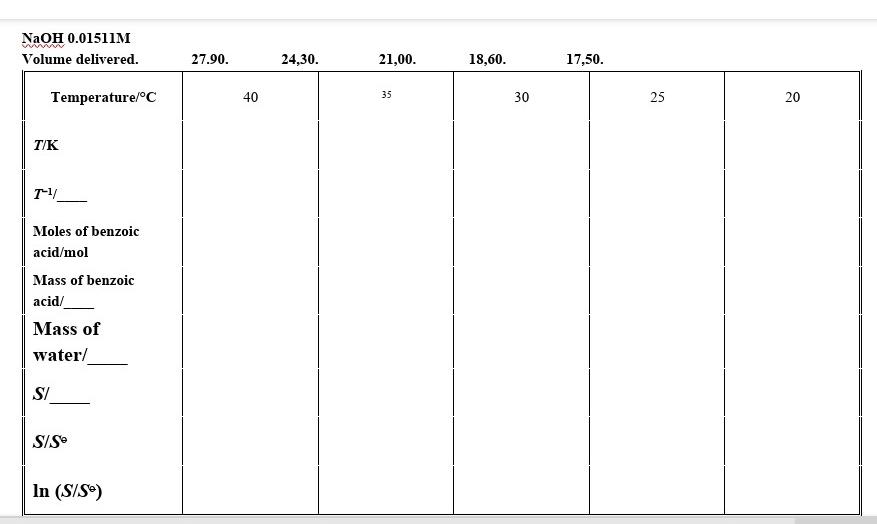 www.chegg.com
www.chegg.com
how to calculate solubility | Chegg.com
Solved b. given ksp calculate solubility example: what is. Solved b. given ksp calculate solubility example: what is. Calculate the solubility of a molecule using ksp values question if a
