Calculating the number of protons, neutrons, and electrons in an atom is a fundamental concept in chemistry. Understanding the composition of atoms is crucial for understanding the properties of elements and how they interact with each other. In this article, we will break down the steps to calculate protons, neutrons, and electrons in an atom, providing you with a comprehensive guide to master this essential skill.
1. Determine the Atomic Number
The atomic number of an element is the number of protons present in the nucleus of an atom. To calculate the number of protons, you need to know the atomic number of the element. The atomic number can be found on the periodic table, and it is usually denoted by the symbol Z. For example, the atomic number of carbon is 6, which means that a carbon atom has 6 protons in its nucleus.
2. Determine the Mass Number
The mass number of an element is the sum of the number of protons and neutrons present in the nucleus of an atom. The mass number is usually denoted by the symbol A. To calculate the number of neutrons, you need to know the mass number and the atomic number. The number of neutrons can be calculated by subtracting the atomic number from the mass number.
3. Calculate the Number of Neutrons
Once you know the mass number and the atomic number, you can calculate the number of neutrons by subtracting the atomic number from the mass number. For example, if the mass number of an atom is 14 and the atomic number is 6, the number of neutrons is 14 - 6 = 8. This means that the atom has 8 neutrons in its nucleus.
4. Determine the Number of Electrons
In a neutral atom, the number of electrons is equal to the number of protons. This means that if you know the atomic number of an element, you can determine the number of electrons. For example, if the atomic number of an element is 6, the number of electrons is also 6.
5. Understand the Concept of Isotopes
Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. This means that isotopes have the same atomic number but different mass numbers. Understanding isotopes is crucial for calculating the number of neutrons in an atom.
6. Apply the Concept of Ionization
Ionization occurs when an atom loses or gains electrons, resulting in the formation of ions. If an atom loses electrons, it becomes a positively charged ion, and if it gains electrons, it becomes a negatively charged ion. Understanding ionization is essential for calculating the number of electrons in an ionized atom.
7. Use the Periodic Table as a Reference
The periodic table is a valuable resource for calculating the number of protons, neutrons, and electrons in an atom. The periodic table provides the atomic number and mass number of each element, which can be used to calculate the number of protons, neutrons, and electrons.
8. Practice with Examples
Practicing with examples is essential for mastering the skill of calculating the number of protons, neutrons, and electrons in an atom. Start with simple examples, such as calculating the number of protons and electrons in a neutral atom, and then move on to more complex examples, such as calculating the number of neutrons in an isotope.
9. Understand the Importance of Accuracy
Accuracy is crucial when calculating the number of protons, neutrons, and electrons in an atom. Small mistakes can lead to incorrect calculations, which can have significant consequences in chemistry and physics. Therefore, it is essential to double-check your calculations and ensure that you are using the correct values for the atomic number and mass number.
10. Review and Reinforce Your Knowledge
Reviewing and reinforcing your knowledge of calculating the number of protons, neutrons, and electrons in an atom is essential for long-term retention and understanding. Regularly reviewing the concepts and practicing with examples will help you to become proficient in calculating the composition of atoms.
If you are looking for How To Calculate The Number of Protons, Neutrons, and Electrons you've visit to the right page. We have 10 Pictures about How To Calculate The Number of Protons, Neutrons, and Electrons like Calculating Protons, Electrons, and Neutrons | PDF, Tutorial 22 Calculation of Number of Protons, Electrons and Neutrons and also Free Printable Protons Neutrons and Electrons Practice Worksheets. Here it is:
How To Calculate The Number Of Protons, Neutrons, And Electrons
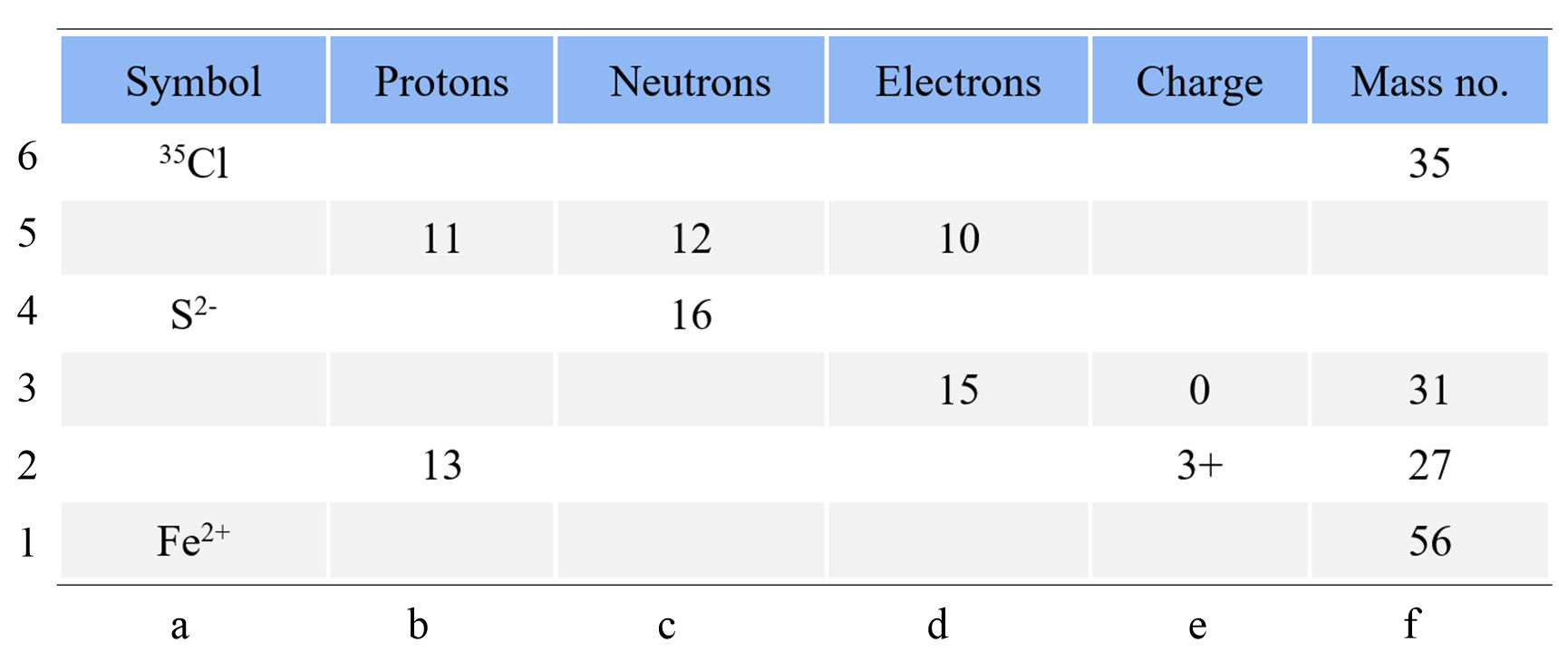 general.chemistrysteps.com
general.chemistrysteps.com
How To Calculate The Number of Protons, Neutrons, and Electrons ...
Calculate The Number Of Protons, Neutrons And Electrons In, 46% OFF
 www.micoope.com.gt
www.micoope.com.gt
Calculate The Number Of Protons, Neutrons And Electrons In, 46% OFF
Calculating Protons, Electrons, And Neutrons | PDF
 www.scribd.com
www.scribd.com
Calculating Protons, Electrons, and Neutrons | PDF
How To Calculate The Number Of Protons, Neutrons, And Electrons
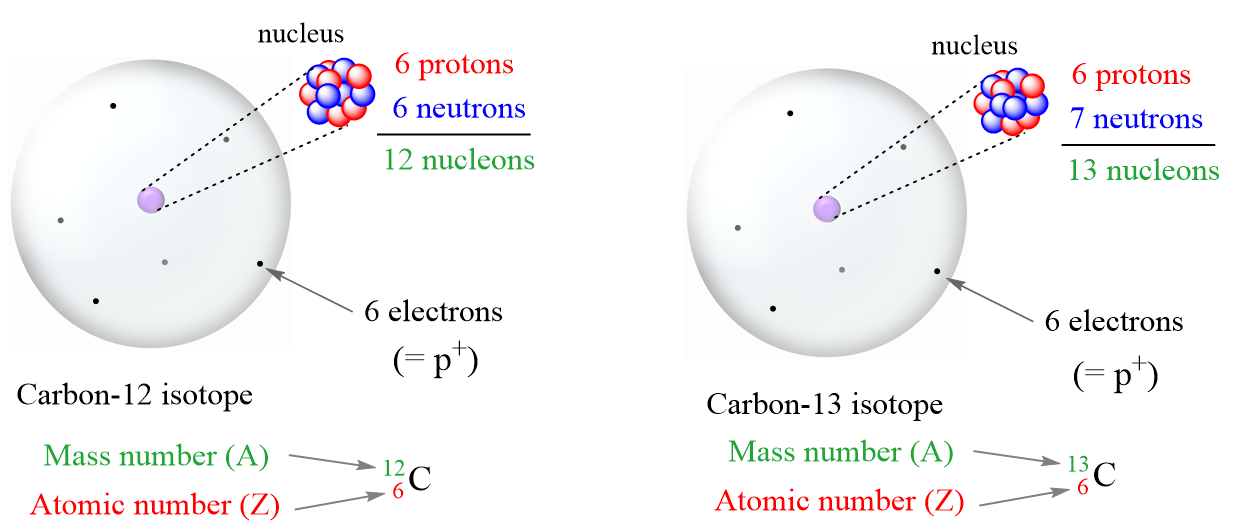 general.chemistrysteps.com
general.chemistrysteps.com
How To Calculate The Number of Protons, Neutrons, and Electrons ...
Free Printable Protons Neutrons And Electrons Practice Worksheets
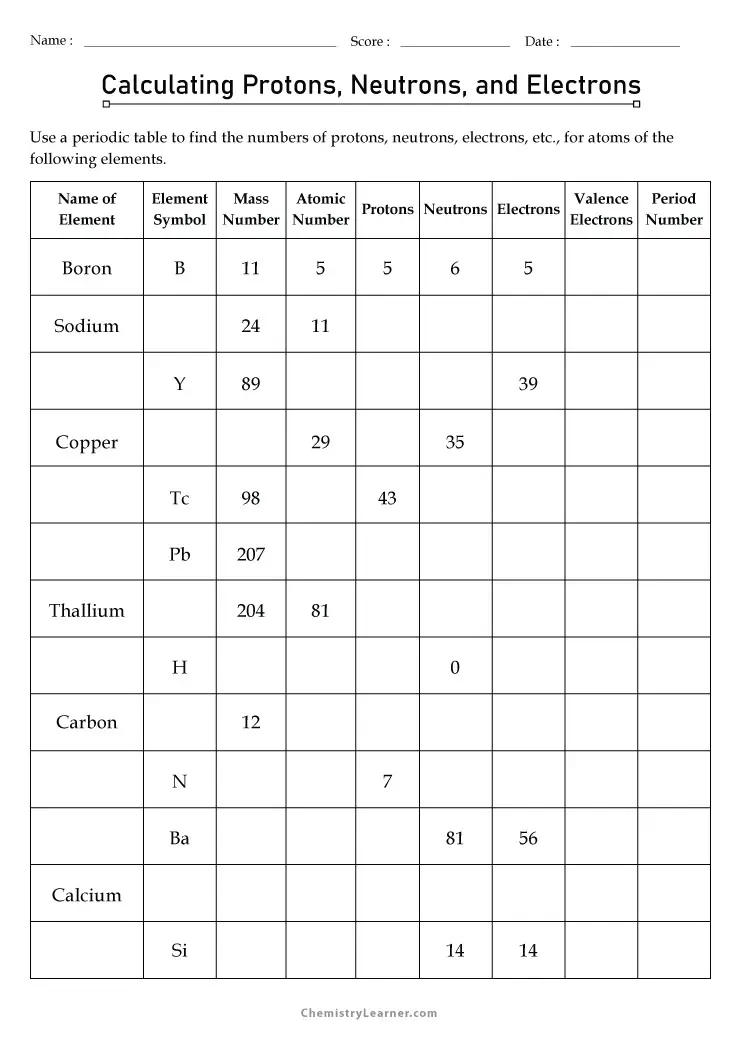 www.chemistrylearner.com
www.chemistrylearner.com
Free Printable Protons Neutrons and Electrons Practice Worksheets
How To Calculate Protons Neutrons And Electrons - Free Worksheets Printable
 lisaworksheets.com
lisaworksheets.com
How To Calculate Protons Neutrons And Electrons - Free Worksheets Printable
Solved: Calculating Protons, Neutrons And Electrons Calculate Protons
Solved: Calculating Protons, Neutrons and Electrons Calculate protons ...
How To Calculate Protons Neutrons And Electrons Worksheet
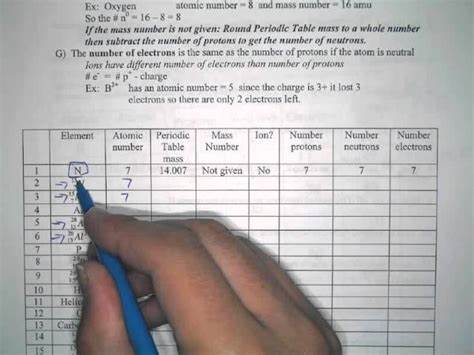 www.primarycalculator.com
www.primarycalculator.com
how to calculate protons neutrons and electrons worksheet ...
Protons Neutrons Electrons Calculator Online
 calculatorshub.net
calculatorshub.net
Protons Neutrons Electrons Calculator Online
Tutorial 22 Calculation Of Number Of Protons, Electrons And Neutrons
 www.scribd.com
www.scribd.com
Tutorial 22 Calculation of Number of Protons, Electrons and Neutrons ...
Solved: calculating protons, neutrons and electrons calculate protons. how to calculate protons neutrons and electrons. calculate the number of protons, neutrons and electrons in, 46% off
