Calculating percent yield is a crucial step in various fields, including chemistry, physics, and engineering. It helps to determine the efficiency of a process or reaction by comparing the actual yield to the theoretical yield. In this article, we will explore the steps to calculate percent yield and provide examples to illustrate the concept.
1. Determine the Theoretical Yield
The first step in calculating percent yield is to determine the theoretical yield of a process or reaction. This can be done by using the stoichiometry of the reaction, which involves calculating the amount of product that can be formed from a given amount of reactants. The theoretical yield is usually expressed in terms of mass or moles.
2. Calculate the Actual Yield
The next step is to calculate the actual yield of the process or reaction. This can be done by measuring the amount of product that is actually formed. The actual yield can be affected by various factors, such as the efficiency of the reaction, the purity of the reactants, and the conditions under which the reaction is carried out.
3. Use the Percent Yield Formula
The percent yield can be calculated using the formula: percent yield = (actual yield / theoretical yield) x 100. This formula allows you to compare the actual yield to the theoretical yield and determine the efficiency of the process or reaction.
4. Plug in the Values
Once you have determined the theoretical yield and the actual yield, you can plug these values into the percent yield formula. For example, if the theoretical yield is 100 grams and the actual yield is 80 grams, you can calculate the percent yield as follows: percent yield = (80 g / 100 g) x 100 = 80%.
5. Consider the Limiting Reactant
In some cases, the reaction may be limited by one of the reactants. In this case, the theoretical yield should be calculated based on the limiting reactant. The limiting reactant is the reactant that is present in the smallest stoichiometric amount.
6. Account for Impurities
Impurities in the reactants or products can affect the actual yield and the percent yield. If the reactants or products are not pure, the actual yield may be lower than expected, which can result in a lower percent yield.
7. Use Significant Figures
When calculating percent yield, it is essential to use significant figures. The number of significant figures in the percent yield should be the same as the number of significant figures in the actual yield and the theoretical yield.
8. Interpret the Results
The percent yield can provide valuable information about the efficiency of a process or reaction. A high percent yield indicates that the process or reaction is efficient, while a low percent yield indicates that there may be problems with the reaction conditions or the purity of the reactants.
9. Optimize the Reaction Conditions
By calculating the percent yield, you can identify areas for improvement in the reaction conditions. For example, if the percent yield is low, you may need to adjust the temperature, pressure, or reaction time to optimize the reaction.
10. Repeat the Calculation
Finally, it is essential to repeat the calculation to ensure that the results are accurate and reliable. By repeating the calculation, you can verify that the percent yield is consistent and that the results are not affected by errors or fluctuations in the reaction conditions.
If you are looking for The Best Way to Calculate Percent Yield in Chemistry - wikiHow you've visit to the right web. We have 10 Pictures about The Best Way to Calculate Percent Yield in Chemistry - wikiHow like The Best Way to Calculate Percent Yield in Chemistry - wikiHow, The Best Way to Calculate Percent Yield in Chemistry - wikiHow and also How to Calculate Percent Yield in Chemistry. Read more:
The Best Way To Calculate Percent Yield In Chemistry - WikiHow
 www.wikihow.com
www.wikihow.com
The Best Way to Calculate Percent Yield in Chemistry - wikiHow
Calculate Theoretical & Percent Yield: Ultimate Guide
 warreninstitute.org
warreninstitute.org
Calculate Theoretical & Percent Yield: Ultimate Guide
Solved 2. Calculate The Theoretical Yield And Percent Yield | Chegg.com
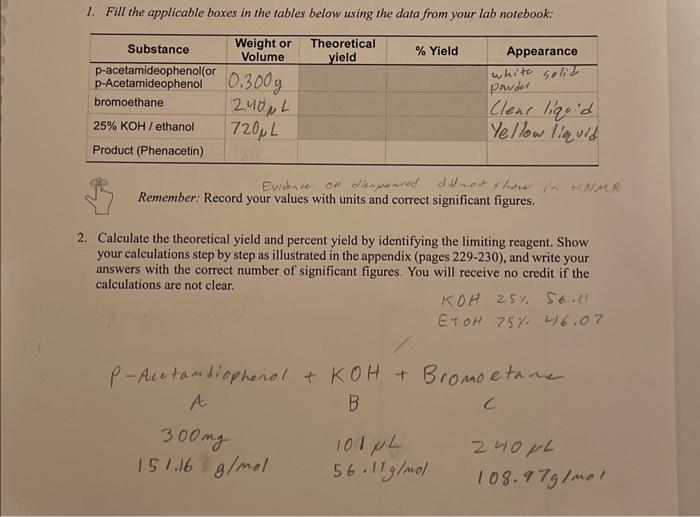 www.chegg.com
www.chegg.com
Solved 2. Calculate the theoretical yield and percent yield | Chegg.com
How To Calculate Percent Yield In Chemistry: 15 Steps
 www.wikihow.com
www.wikihow.com
How to Calculate Percent Yield in Chemistry: 15 Steps
How To Calculate Percent Yield In Chemistry: 15 Steps
 www.wikihow.com
www.wikihow.com
How to Calculate Percent Yield in Chemistry: 15 Steps
How Do You Calculate Percent Yield In Organic Synthesis?
 scienceoxygen.com
scienceoxygen.com
How do you calculate percent yield in organic synthesis?
How To Calculate Percent Yield In Chemistry
 www.24houranswers.com
www.24houranswers.com
How to Calculate Percent Yield in Chemistry
How To Calculate Theoretical Yield: 12 Steps (with Pictures)
 www.wikihow.com
www.wikihow.com
How to Calculate Theoretical Yield: 12 Steps (with Pictures)
How To Calculate Percent Yield In Chemistry: 13 Steps
 www.wikihow.com
www.wikihow.com
How to Calculate Percent Yield in Chemistry: 13 Steps
The Best Way To Calculate Percent Yield In Chemistry - WikiHow
 www.wikihow.com
www.wikihow.com
The Best Way to Calculate Percent Yield in Chemistry - wikiHow
how to calculate percent yield in chemistry: 15 steps. How to calculate percent yield in chemistry: 15 steps. Solved 2. calculate the theoretical yield and percent yield
