Calculating partial pressure is a crucial concept in chemistry, particularly when dealing with gases. Whether you're a student or a professional, understanding how to calculate partial pressure can help you grasp the behavior of gases in various mixtures. In this article, we'll break down the steps to calculate partial pressure, making it easier for you to tackle problems with confidence. So, let's dive in and explore the key points to keep in mind when calculating partial pressure.
1. Understand the Concept of Partial Pressure
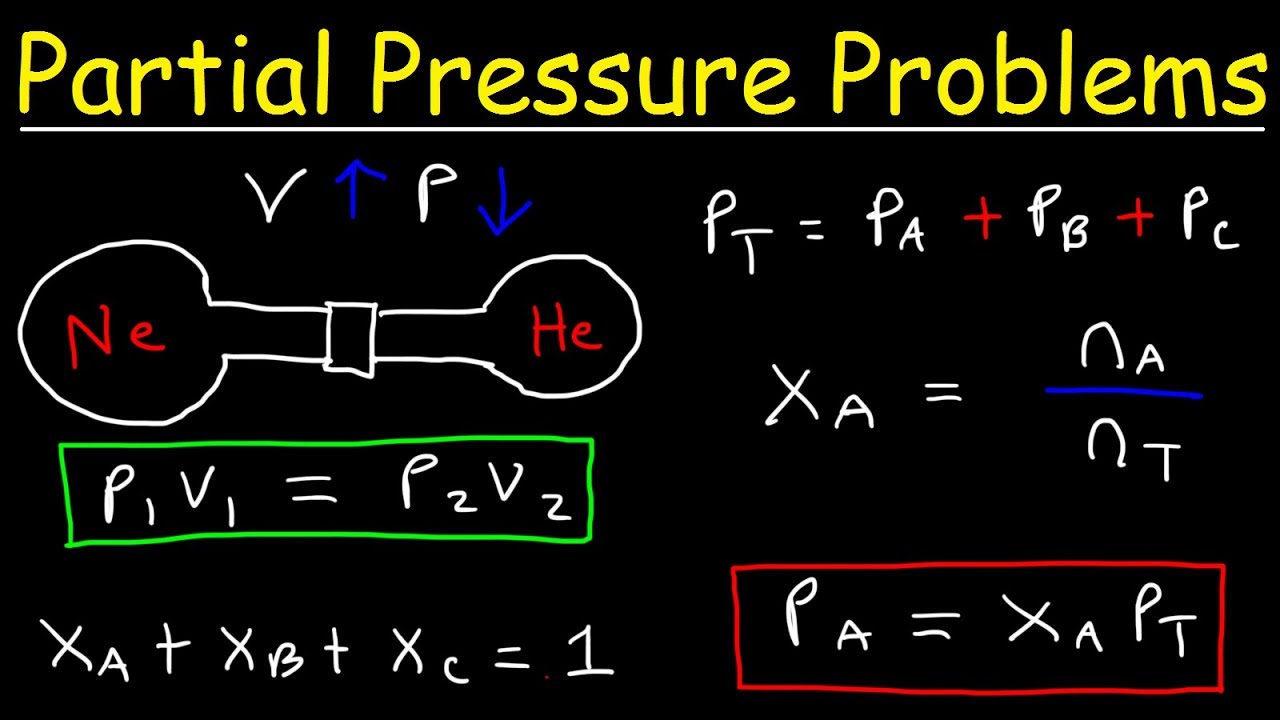
The concept of partial pressure is based on the idea that each gas in a mixture exerts its own pressure, which is independent of the other gases present. This pressure is proportional to the number of moles of the gas and is a fundamental principle in understanding gas behavior. To calculate partial pressure, you need to know the total pressure of the mixture and the mole fraction of the gas you're interested in.
2. Identify the Total Pressure of the Mixture
The total pressure of the mixture is the sum of the partial pressures of all the gases present. This value is usually given in the problem or can be measured using a pressure gauge. Make sure to note the units of pressure, as they can vary (e.g., atmospheres, pascals, or millimeters of mercury).
3. Determine the Mole Fraction of the Gas
The mole fraction of a gas is the ratio of the number of moles of that gas to the total number of moles of all gases in the mixture. You can calculate the mole fraction by dividing the number of moles of the gas by the total number of moles. This value is crucial in calculating the partial pressure of the gas.
4. Use the Formula for Partial Pressure
The formula for partial pressure (P) is given by P = X * P_total, where X is the mole fraction of the gas and P_total is the total pressure of the mixture. By plugging in the values, you can calculate the partial pressure of the gas. Make sure to double-check your units and calculations to ensure accuracy.
5. Apply the Ideal Gas Law (Optional)
In some cases, you may need to use the ideal gas law (PV = nRT) to calculate the partial pressure of a gas. This law relates the pressure, volume, and temperature of a gas to the number of moles. By rearranging the equation, you can solve for partial pressure if you know the other variables.
6. Consider the Effects of Temperature and Volume
Temperature and volume can affect the partial pressure of a gas. As temperature increases, the molecules of the gas gain kinetic energy and move more rapidly, resulting in a higher partial pressure. Similarly, an increase in volume can lead to a decrease in partial pressure. Keep these factors in mind when calculating partial pressure.
7. Be Mindful of Gas Interactions and Reactions
In a mixture, gases can interact with each other through chemical reactions or physical interactions. These interactions can affect the partial pressure of the gases involved. Be aware of any potential reactions or interactions that may impact your calculation.
8. Check Your Units and Calculations
Finally, it's essential to verify that your units are consistent and your calculations are accurate. Partial pressure calculations involve several steps, and a small mistake can lead to a significant error. Take the time to review your work and ensure that your answer makes sense in the context of the problem.
9. Practice with Real-World Examples
To become proficient in calculating partial pressure, practice with real-world examples or problems. This will help you develop a deeper understanding of the concept and improve your ability to apply the formula in different scenarios.
10. Review and Refine Your Understanding
Calculating partial pressure is a skill that requires practice and review. As you work through problems and examples, refine your understanding of the concept and the formulas involved. With time and practice, you'll become more confident and proficient in calculating partial pressure, making it easier to tackle complex chemistry problems.
If you are looking for Partial Pressure- Formula, Dalton's Law, Mixture of Ideal Gas, Examples you've visit to the right place. We have 10 Pictures about Partial Pressure- Formula, Dalton's Law, Mixture of Ideal Gas, Examples like How to Calculate Partial Pressure: Step-by-Step Solution, How to Calculate Partial Pressure: Step-by-Step Solution and also How do you calculate partial pressure of co2?. Here it is:
Partial Pressure- Formula, Dalton's Law, Mixture Of Ideal Gas, Examples
 byjus.com
byjus.com
Partial Pressure- Formula, Dalton's Law, Mixture of Ideal Gas, Examples ...
How To Calculate Partial Pressure: Step-by-Step Solution
 www.wikihow.com
www.wikihow.com
How to Calculate Partial Pressure: Step-by-Step Solution
How To Calculate Partial Pressure - EdithtinDean
 edithtindean.blogspot.com
edithtindean.blogspot.com
How to Calculate Partial Pressure - EdithtinDean
SOLVED: Calculate Pressure Using Dalton's Law Of Partial Pressures
 www.numerade.com
www.numerade.com
SOLVED: Calculate pressure using Dalton's law of partial pressures ...
Partial Pressure- Formula, Dalton's Law, Mixture Of Ideal Gas, Examples
 byjus.com
byjus.com
Partial Pressure- Formula, Dalton's Law, Mixture of Ideal Gas, Examples ...
How To Calculate Partial Pressure: Step-by-Step Solution
 www.wikihow.com
www.wikihow.com
How to Calculate Partial Pressure: Step-by-Step Solution
How Do You Calculate Partial Pressure Of Co2?
 scienceoxygen.com
scienceoxygen.com
How do you calculate partial pressure of co2?
How To Calculate Partial Pressure
 jamari-bogspotchapman.blogspot.com
jamari-bogspotchapman.blogspot.com
How to Calculate Partial Pressure
Partial Pressure Calculation
 mungfali.com
mungfali.com
Partial Pressure Calculation
How To Calculate Partial Pressure: Step-by-Step Solution
 www.wikihow.com
www.wikihow.com
How to Calculate Partial Pressure: Step-by-Step Solution
How to calculate partial pressure. how to calculate partial pressure: step-by-step solution. how to calculate partial pressure: step-by-step solution
