In the realm of chemistry, the solubility product constant (Ksp) is a crucial parameter that helps us understand the equilibrium between a solid ionic compound and its ions in a solution. For students and chemists alike, calculating Ksp is an essential skill to master. In this article, we will delve into the world of Ksp calculations, exploring the steps and considerations necessary to determine this vital constant. From understanding the concept of Ksp to applying it in real-world scenarios, we will break down the process into manageable, bite-sized chunks.
1. Write Down the Dissociation Reaction
The first step in calculating Ksp is to write down the dissociation reaction of the ionic compound. This involves breaking down the compound into its constituent ions and understanding how they interact with each other in a solution. For example, the dissociation reaction of calcium carbonate (CaCO3) can be written as CaCO3 (s) ⇌ Ca2+ (aq) + CO32- (aq). This step is crucial in identifying the ions involved and their respective concentrations.
2. Identify the Ions and Their Concentrations
Once the dissociation reaction is written down, the next step is to identify the ions involved and their concentrations. This can be done by looking at the balanced chemical equation and determining the stoichiometry of the reaction. For instance, in the dissociation reaction of CaCO3, the concentration of Ca2+ ions is equal to the concentration of CO32- ions. Understanding the concentrations of the ions is vital in calculating Ksp.
3. Use the Ksp Formula
The Ksp formula is given by Ksp = [A]^a [B]^b, where [A] and [B] are the concentrations of the ions, and a and b are their respective stoichiometric coefficients. For example, in the dissociation reaction of CaCO3, the Ksp formula would be Ksp = [Ca2+][CO32-]. This formula is used to calculate the Ksp value, which represents the equilibrium between the solid and the ions in the solution.
4. Determine the Concentrations of the Ions
To calculate Ksp, the concentrations of the ions must be determined. This can be done by using various methods such as titration, spectroscopy, or chromatography. The concentrations of the ions can also be calculated using the stoichiometry of the reaction. For instance, if the concentration of Ca2+ ions is 0.1 M, the concentration of CO32- ions would also be 0.1 M, as they are present in a 1:1 ratio.
5. Plug in the Values and Calculate Ksp
Once the concentrations of the ions are determined, the Ksp value can be calculated by plugging in the values into the Ksp formula. For example, if the concentration of Ca2+ ions is 0.1 M and the concentration of CO32- ions is 0.1 M, the Ksp value would be Ksp = [0.1][0.1] = 0.01. This represents the equilibrium constant between the solid CaCO3 and its ions in the solution.
6. Consider the Common Ion Effect
The common ion effect is a phenomenon that occurs when a solution contains two or more ions that are common to multiple compounds. This can affect the Ksp calculation, as the concentration of the common ion can be influenced by the presence of other compounds. For instance, if a solution contains Ca2+ ions from multiple sources, the concentration of Ca2+ ions must be taken into account when calculating Ksp.
7. Consider the pH of the Solution
The pH of the solution can also affect the Ksp calculation, as certain ions can react with hydrogen or hydroxide ions to form new compounds. For example, in a basic solution, the CO32- ion can react with hydrogen ions to form HCO3- ions, which can affect the Ksp calculation. Therefore, it is essential to consider the pH of the solution when calculating Ksp.
8. Use Ksp Values to Predict Solubility
Once the Ksp value is calculated, it can be used to predict the solubility of the ionic compound. A high Ksp value indicates that the compound is highly soluble, while a low Ksp value indicates that the compound is less soluble. For instance, if the Ksp value of CaCO3 is 0.01, it indicates that CaCO3 is relatively insoluble in water.
9. Consider the Temperature Dependence of Ksp
Ksp values are temperature-dependent, meaning that they can change with variations in temperature. This is because the equilibrium constant is affected by the temperature of the solution. For example, the Ksp value of CaCO3 can increase with increasing temperature, indicating that CaCO3 becomes more soluble at higher temperatures.
10. Practice and Review Ksp Calculations
Finally, it is essential to practice and review Ksp calculations to become proficient in determining this vital constant. By working through numerous examples and problems, students and chemists can develop a deeper understanding of the concept of Ksp and its applications in chemistry. With practice and review, calculating Ksp becomes a straightforward process that can be applied to a wide range of chemical compounds and reactions.
If you are looking for Solved given-Use the Ksp values to calculate the molar | Chegg.com you've came to the right web. We have 10 Pictures about Solved given-Use the Ksp values to calculate the molar | Chegg.com like KSP 6 | PDF | Solubility | Homogeneous Chemical Mixtures, Calculate Ksp: Solubility Product Constant Calculation Guide and also Calculate Ksp: Solubility Product Constant Calculation Guide. Here you go:
Solved Given-Use The Ksp Values To Calculate The Molar | Chegg.com
 www.chegg.com
www.chegg.com
Solved given-Use the Ksp values to calculate the molar | Chegg.com
How Do You Calculate Ksp From Solubility?
 scienceoxygen.com
scienceoxygen.com
How do you calculate Ksp from solubility?
Solved Solubility Product, Ksp - Worksheet 2 1. Calculate | Chegg.com
 worksheets.clipart-library.com
worksheets.clipart-library.com
Solved Solubility Product, Ksp - Worksheet 2 1. Calculate | Chegg.com ...
Solved Refer To The Ksp Values To Calculate The Molar | Chegg.com
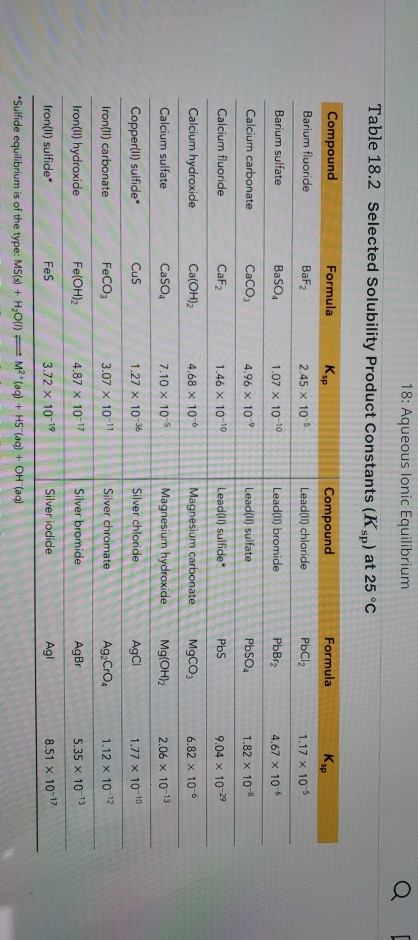 www.chegg.com
www.chegg.com
Solved refer to the Ksp values to calculate the molar | Chegg.com
How To Calculate The Molar Solubility From Ksp Values??? - WizEdu
 wizedu.com
wizedu.com
how to calculate the molar solubility from Ksp values??? - WizEdu
KSP 6 | PDF | Solubility | Homogeneous Chemical Mixtures
 www.scribd.com
www.scribd.com
KSP 6 | PDF | Solubility | Homogeneous Chemical Mixtures
Solved Use The K_sp Values In The Table To Calculate The | Chegg.com
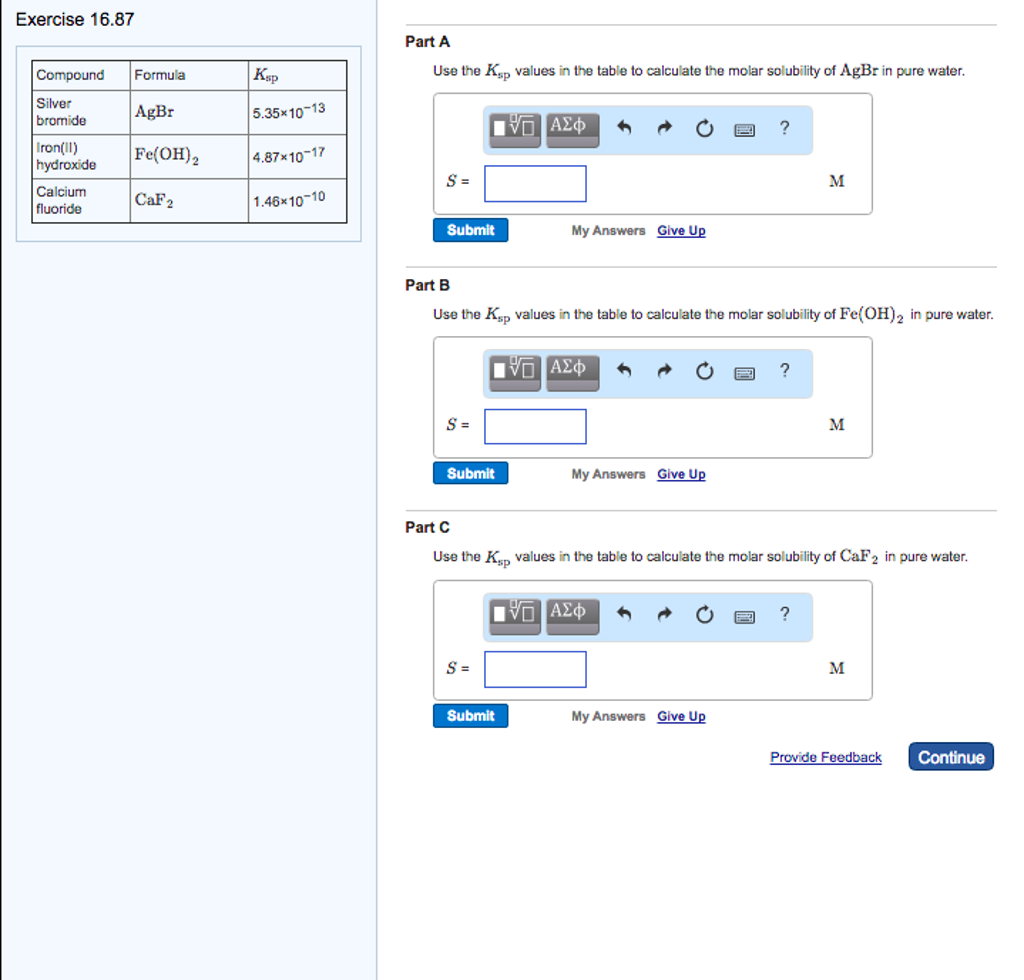 www.chegg.com
www.chegg.com
Solved Use the K_sp values in the table to calculate the | Chegg.com
Calculate Ksp: Solubility Product Constant Calculation Guide
 sourcetable.com
sourcetable.com
Calculate Ksp: Solubility Product Constant Calculation Guide
Calculate Ksp: Solubility Product Constant Calculation Guide
 sourcetable.com
sourcetable.com
Calculate Ksp: Solubility Product Constant Calculation Guide
Molar Solubility To Ksp Calculator, Chemistry
 topblogtenz.com
topblogtenz.com
Molar solubility to Ksp Calculator, Chemistry
Solved given-use the ksp values to calculate the molar. Solved given-use the ksp values to calculate the molar. How do you calculate ksp from solubility?
