To calculate the equilibrium constant of a chemical reaction, it is essential to understand the concept of equilibrium and the factors that affect it. The equilibrium constant, denoted by K, is a mathematical expression that describes the ratio of the concentrations of the products to the concentrations of the reactants at equilibrium. In this article, we will delve into the steps to calculate the equilibrium constant, providing a comprehensive guide for chemists and researchers.
1. Write Down the Balanced Chemical Equation
The first step in calculating the equilibrium constant is to write down the balanced chemical equation for the reaction. This equation should include all the reactants and products, with their respective stoichiometric coefficients. The balanced equation will help you identify the concentrations of the reactants and products, which are necessary for calculating the equilibrium constant.
2. Identify the Concentrations of the Reactants and Products
Once you have the balanced chemical equation, identify the concentrations of the reactants and products. These concentrations can be expressed in terms of molarity (M), which is defined as the number of moles of a substance per liter of solution. The concentrations of the reactants and products can be measured experimentally or obtained from literature values.
3. Determine the Expression for the Equilibrium Constant
The expression for the equilibrium constant, K, is determined by the balanced chemical equation. For a general reaction of the form aA + bB ⇌ cC + dD, the equilibrium constant expression is K = [C]^c [D]^d / [A]^a [B]^b, where [A], [B], [C], and [D] are the concentrations of the reactants and products.
4. Calculate the Concentrations of the Reactants and Products at Equilibrium
To calculate the equilibrium constant, you need to know the concentrations of the reactants and products at equilibrium. These concentrations can be calculated using the initial concentrations of the reactants and the stoichiometric coefficients of the balanced equation. The equilibrium concentrations can also be measured experimentally using various analytical techniques.
5. Plug in the Values into the Equilibrium Constant Expression
Once you have the concentrations of the reactants and products at equilibrium, plug these values into the equilibrium constant expression. Make sure to use the correct units and significant figures when calculating the equilibrium constant.
6. Calculate the Equilibrium Constant
With the values plugged into the equilibrium constant expression, calculate the equilibrium constant. This can be done using a calculator or software program, such as a spreadsheet or a programming language. The calculated value of the equilibrium constant should be a unitless quantity.
7. Check the Units and Significant Figures
After calculating the equilibrium constant, check the units and significant figures to ensure that they are correct. The equilibrium constant should be a unitless quantity, and the significant figures should be consistent with the experimental measurements or literature values.
8. Compare the Calculated Value with Literature Values
Finally, compare the calculated value of the equilibrium constant with literature values to ensure that it is consistent with established values. This can help you verify the accuracy of your calculation and identify any potential errors or discrepancies.
9. Consider the Limitations and Assumptions
When calculating the equilibrium constant, consider the limitations and assumptions of the calculation. The equilibrium constant is a mathematical expression that assumes ideal behavior and neglects non-ideal effects, such as activity coefficients and ion pairing. These limitations should be taken into account when interpreting the results.
10. Document the Calculation and Results
Finally, document the calculation and results, including the balanced chemical equation, the concentrations of the reactants and products, the equilibrium constant expression, and the calculated value of the equilibrium constant. This documentation will help you track your work and provide a clear record of your calculation for future reference.
If you are searching about Equilibrium Constant Calculator you've came to the right web. We have 10 Pics about Equilibrium Constant Calculator like Equilibrium Constant Calculator, How to calculate equilibrium constant using ice table and also Solved EXPERIMENTAL EQUILIBRIUM CONSTANTS Now, use ICE | Chegg.com. Here you go:
Equilibrium Constant Calculator
 www.examples.com
www.examples.com
Equilibrium Constant Calculator
Learn How To Calculate An Equilibrium Constant Kc. | Teaching Chemistry
 www.pinterest.co.uk
www.pinterest.co.uk
Learn how to calculate an equilibrium constant Kc. | Teaching chemistry ...
How To Calculate Equilibrium Constant Using Ice Table
 answeringeverything.com
answeringeverything.com
How to calculate equilibrium constant using ice table ...
Using The Following Data, Calculate Equilibrium Constant K For The Reacti..
 askfilo.com
askfilo.com
Using the following data, calculate equilibrium constant K for the reacti..
Solved EXPERIMENTAL EQUILIBRIUM CONSTANTS Now, Use ICE | Chegg.com
 www.chegg.com
www.chegg.com
Solved EXPERIMENTAL EQUILIBRIUM CONSTANTS Now, use ICE | Chegg.com
Solved Calculate The Equilibrium Constant For The Chemical | Chegg.com
 www.chegg.com
www.chegg.com
Solved Calculate the equilibrium constant for the chemical | Chegg.com
Solved EXPERIMENTAL EQUILIBRIUM CONSTANTS Now, Use ICE | Chegg.com
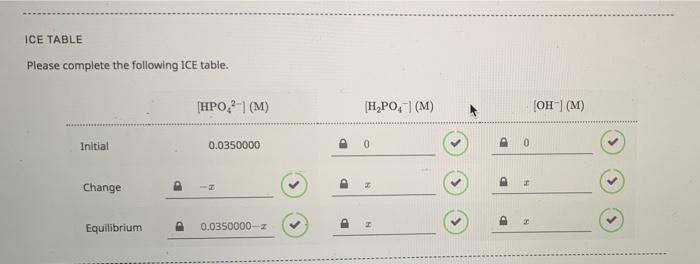 www.chegg.com
www.chegg.com
Solved EXPERIMENTAL EQUILIBRIUM CONSTANTS Now, use ICE | Chegg.com
How To Calculate Equilibrium Constant From E Cell At Marcia Donald Blog
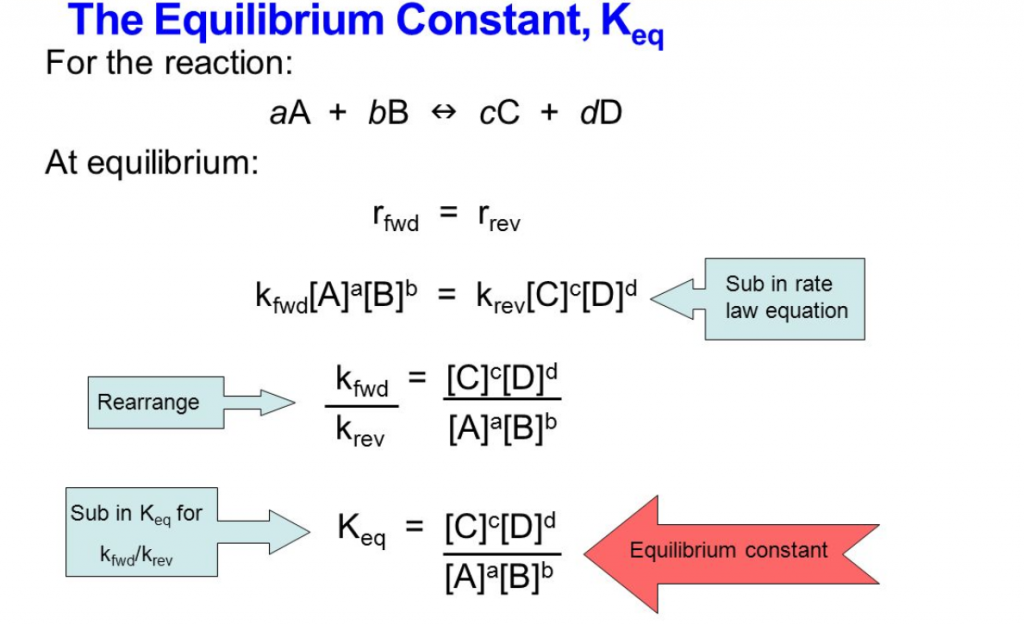 storage.googleapis.com
storage.googleapis.com
How To Calculate Equilibrium Constant From E Cell at Marcia Donald blog
SOLVED:Calculate The Equilibrium Constant For Each Of The Reactions In
 www.numerade.com
www.numerade.com
SOLVED:Calculate the equilibrium constant for each of the reactions in ...
Calculate The Equilibrium Constant For The Reaction Given Below At 400 K..
 askfilo.com
askfilo.com
Calculate the equilibrium constant for the reaction given below at 400 K..
Solved experimental equilibrium constants now, use ice. Using the following data, calculate equilibrium constant k for the reacti... Calculate the equilibrium constant for the reaction given below at 400 k..
