Calculating electrons can seem like a daunting task, especially for those who are new to the world of chemistry and physics. However, with the right approach and a few simple steps, you can master the art of electron calculation. In this article, we'll take you through the process of calculating electrons in a clear and concise manner, making it easy for you to understand and apply the concepts to your studies or work.
1. Understand the Atomic Structure
The first step to calculating electrons is to understand the atomic structure. Atoms are the building blocks of matter, and they consist of protons, neutrons, and electrons. Protons and neutrons are found in the nucleus, while electrons orbit around the nucleus. The number of protons in an atom determines the element of an atom, and the number of electrons in a neutral atom is equal to the number of protons.
2. Determine the Number of Protons
To calculate the number of electrons in an atom, you need to determine the number of protons. The number of protons in an atom is also known as the atomic number. You can find the atomic number of an element by looking at the periodic table. The atomic number is usually represented by the symbol Z.
3. Determine the Number of Electrons in a Neutral Atom
In a neutral atom, the number of electrons is equal to the number of protons. This means that if you know the atomic number of an element, you can determine the number of electrons in a neutral atom. For example, if the atomic number of an element is 6, then the number of electrons in a neutral atom of that element is also 6.
4. Calculate the Number of Electrons in an Ion
Ions are atoms or molecules that have gained or lost electrons, resulting in a net positive or negative charge. To calculate the number of electrons in an ion, you need to know the charge of the ion. If the ion has a positive charge, it has lost electrons, and if it has a negative charge, it has gained electrons. You can calculate the number of electrons in an ion by adding or subtracting the charge from the number of electrons in a neutral atom.
5. Understand Electron Shells
Electron shells are the regions around the nucleus where electrons are found. Each shell has a specific capacity for electrons, and the electrons in each shell are arranged in a specific pattern. Understanding electron shells is important for calculating the number of electrons in an atom, as it helps you to determine the arrangement of electrons in the atom.
6. Determine the Number of Valence Electrons
Valence electrons are the electrons in the outermost shell of an atom. These electrons are involved in chemical bonding and determine the chemical properties of an element. To calculate the number of valence electrons, you need to know the electron configuration of the atom, which can be determined using the periodic table.
7. Calculate the Number of Core Electrons
Core electrons are the electrons in the inner shells of an atom. These electrons are not involved in chemical bonding and do not affect the chemical properties of an element. To calculate the number of core electrons, you need to subtract the number of valence electrons from the total number of electrons in the atom.
8. Use the Periodic Table to Calculate Electrons
The periodic table is a powerful tool for calculating electrons. By looking at the periodic table, you can determine the atomic number, electron configuration, and number of valence electrons for a particular element. You can use this information to calculate the total number of electrons in an atom or ion.
9. Apply the Octet Rule
The octet rule states that atoms tend to gain, lose, or share electrons to achieve a full outer shell with eight electrons. This rule can be used to calculate the number of electrons in an ion or molecule. By applying the octet rule, you can determine the number of electrons that are gained or lost by an atom to achieve a stable electronic configuration.
10. Practice with Examples
Calculating electrons can seem complex, but with practice, you can become proficient. Try practicing with examples to calculate the number of electrons in different atoms and ions. This will help you to apply the concepts and formulas learned in this article and develop a deeper understanding of electron calculation.
If you are searching about Calculate Protons Electrons and Neutrons by Marine Science Store you've came to the right place. We have 10 Pics about Calculate Protons Electrons and Neutrons by Marine Science Store like Calculating Protons, Electrons, and Neutrons | PDF, valenceelectrons.com - Your Ultimate Guide to the Periodic Table and also Calculating Protons, Electrons, and Neutrons | PDF. Here you go:
Calculate Protons Electrons And Neutrons By Marine Science Store
 www.teacherspayteachers.com
www.teacherspayteachers.com
Calculate Protons Electrons and Neutrons by Marine Science Store
How To Find Electrons: 6 Steps (with Pictures) - WikiHow
 www.wikihow.com
www.wikihow.com
How to Find Electrons: 6 Steps (with Pictures) - wikiHow
How To Find Electrons: 6 Steps (with Pictures) - WikiHow
 www.wikihow.com
www.wikihow.com
How to Find Electrons: 6 Steps (with Pictures) - wikiHow
Valenceelectrons.com - Your Ultimate Guide To The Periodic Table
 valenceelectrons.com
valenceelectrons.com
valenceelectrons.com - Your Ultimate Guide to the Periodic Table
How To Find Electrons: 6 Steps (with Pictures) - WikiHow
 www.wikihow.com
www.wikihow.com
How to Find Electrons: 6 Steps (with Pictures) - wikiHow
Calculating Protons, Electrons, And Neutrons | PDF
 www.scribd.com
www.scribd.com
Calculating Protons, Electrons, and Neutrons | PDF
How To Find Electrons: 7 Steps (with Pictures) - WikiHow
 www.wikihow.com
www.wikihow.com
How to Find Electrons: 7 Steps (with Pictures) - wikiHow
How To Find Electrons: 6 Steps (with Pictures) - WikiHow
 www.wikihow.com
www.wikihow.com
How to Find Electrons: 6 Steps (with Pictures) - wikiHow
How To Find Electrons: 6 Steps (with Pictures) - WikiHow
 www.wikihow.com
www.wikihow.com
How to Find Electrons: 6 Steps (with Pictures) - wikiHow
How To Calculate The Number Of Protons, Neutrons, And Electrons
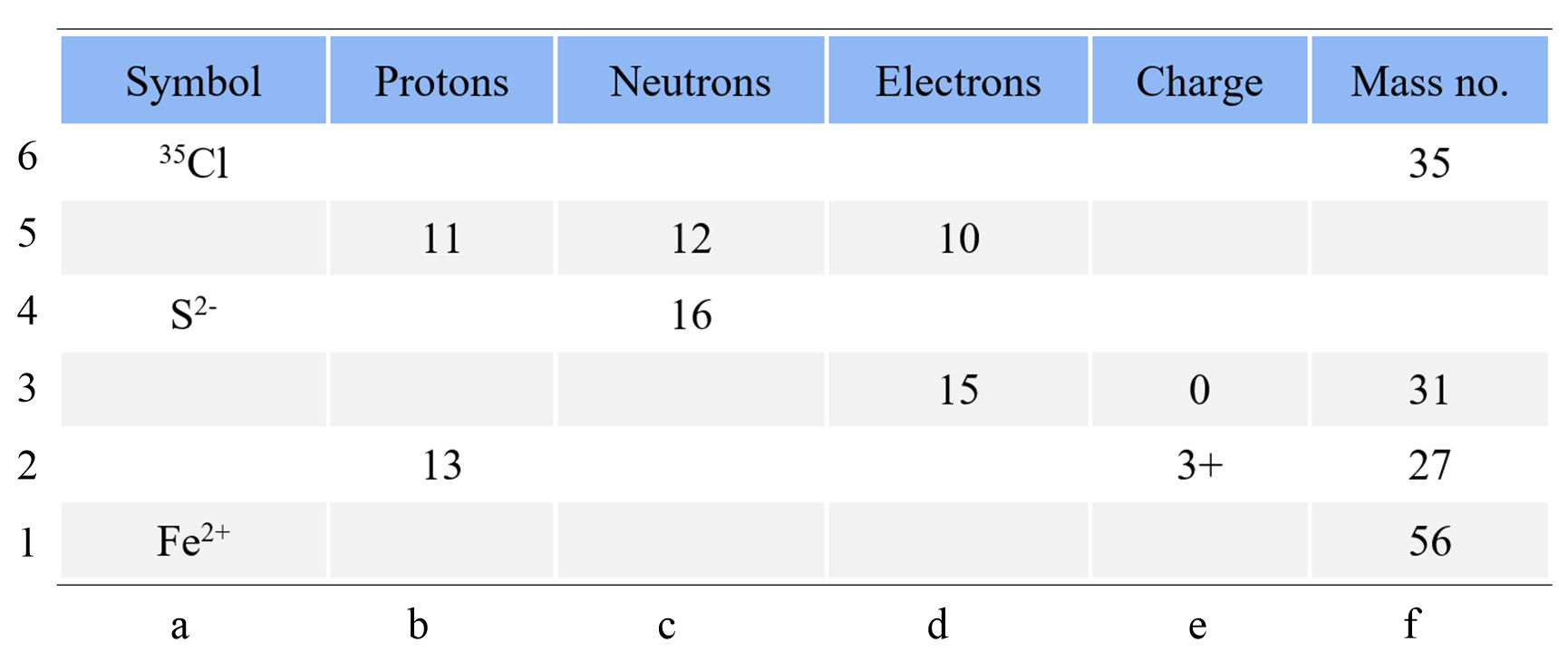 general.chemistrysteps.com
general.chemistrysteps.com
How To Calculate The Number of Protons, Neutrons, and Electrons ...
How to find electrons: 7 steps (with pictures). Valenceelectrons.com. How to find electrons: 6 steps (with pictures)
