Are you ready to dive into the wonderful world of thermodynamics and calculate delta H like a pro? Delta H, also known as the enthalpy change, is a fundamental concept in chemistry that can be a bit tricky to wrap your head around. But don't worry, we've got you covered. In this listicle, we'll break down the steps to calculate delta H in a way that's easy to understand and (dare we say it) fun. So, grab your calculator, put on your thinking cap, and let's get started!
1. Understand the Concept of Enthalpy
Before we dive into the calculation, it's essential to understand what enthalpy is. Enthalpy is a measure of the total energy of a system, including the internal energy and the energy associated with the pressure and volume of a system. Think of it like your bank account - it's the total amount of money you have, including the cash in your wallet and the money in your savings account.
2. Identify the Type of Reaction
Not all reactions are created equal, and the type of reaction you're dealing with will affect how you calculate delta H. Are you working with a combustion reaction, a synthesis reaction, or something else entirely? Make sure you know the type of reaction you're dealing with before you start calculating.
3. Gather Your Data
To calculate delta H, you'll need to know the initial and final states of the system, including the temperature, pressure, and volume. You'll also need to know the specific heat capacity of the substances involved and the amount of heat transferred during the reaction. It's like gathering ingredients for a recipe - you need to have all the right stuff before you can start cooking.
4. Choose the Right Equation
There are several equations you can use to calculate delta H, depending on the type of reaction and the data you have available. The most common equation is delta H = Q - P * delta V, where Q is the heat transferred, P is the pressure, and delta V is the change in volume. But don't worry if that looks like gibberish - we'll break it down step by step.
5. Plug in the Values
Now it's time to plug in the values you've gathered into the equation. Make sure you're using the correct units and that you're accounting for any signs (positive or negative) that might affect the calculation. It's like solving a puzzle - you need to make sure all the pieces fit together just right.
6. Consider the Signs
When calculating delta H, it's essential to consider the signs of the values you're using. Is the reaction exothermic (releasing heat) or endothermic (absorbing heat)? The sign of the heat transferred will affect the sign of delta H, so make sure you're getting it right.
7. Check Your Units
Thermodynamics can be a real unit-fest, with joules, calories, and kilojoules all making an appearance. Make sure you're using the correct units for each value and that you're converting between units correctly. It's like cooking a recipe - if you use the wrong units, you might end up with a dish that's inedible.
8. Calculate the Delta H
Finally, it's time to calculate the delta H. Plug in the values, consider the signs, and check your units. If you've done everything correctly, you should get a value for delta H that makes sense. And if you don't, don't worry - just go back and check your work.
9. Interpret the Results
So you've calculated the delta H - now what? The value of delta H can tell you a lot about the reaction, including whether it's spontaneous or not. A negative delta H indicates a spontaneous reaction, while a positive delta H indicates a non-spontaneous reaction. It's like getting the results of a test - you need to understand what the results mean in order to make sense of them.
10. Practice, Practice, Practice
Calculating delta H is like riding a bike - it takes practice to get it right. The more you practice, the more comfortable you'll become with the equations and the concepts. So don't be afraid to try out a few practice problems and see how you do. With a little practice, you'll be a delta H master in no time.
If you are searching about Solved Question: 1 Calculate delta H for the reaction | Chegg.com you've came to the right web. We have 10 Images about Solved Question: 1 Calculate delta H for the reaction | Chegg.com like How to calculate Delta H? - Problems, Formulas, Units, How do you calculate delta H in chemistry? and also Solved Part A: calculate delta H for the reaction between | Chegg.com. Read more:
Solved Question: 1 Calculate Delta H For The Reaction | Chegg.com
 www.chegg.com
www.chegg.com
Solved Question: 1 Calculate delta H for the reaction | Chegg.com
How To Calculate Delta H? - Problems, Formulas, Units
 topblogtenz.com
topblogtenz.com
How to calculate Delta H? - Problems, Formulas, Units
How Do You Calculate Delta H In Chemistry?
 scienceoxygen.com
scienceoxygen.com
How do you calculate delta H in chemistry?
Solved Part A: Calculate Delta H For The Reaction Between | Chegg.com
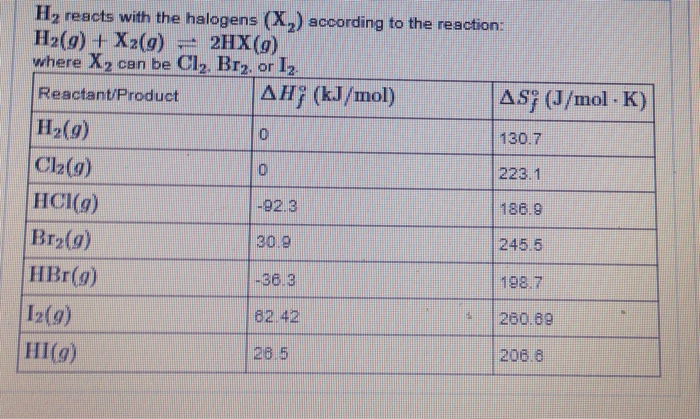 www.chegg.com
www.chegg.com
Solved Part A: calculate delta H for the reaction between | Chegg.com
Solved Using The ΔH° given In The Table, Calculate The | Chegg.com
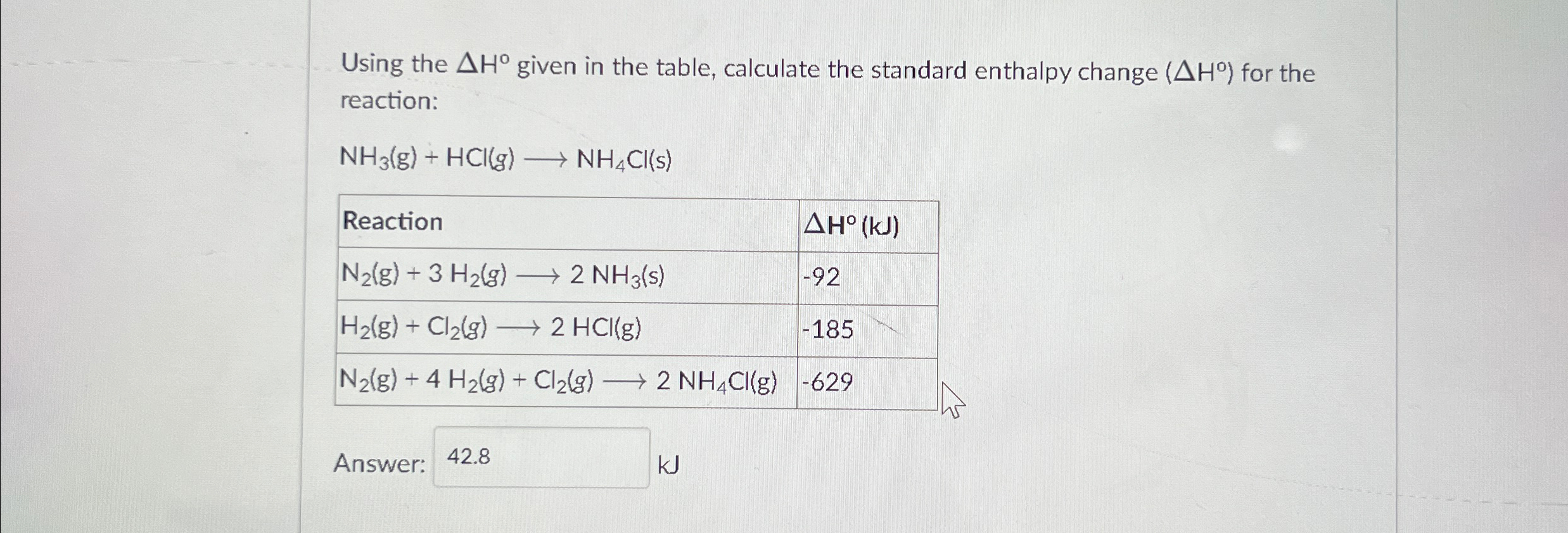 www.chegg.com
www.chegg.com
Solved Using the ΔH° given in the table, calculate the | Chegg.com
How To Calculate Delta H Of Neutralization
 allanfersmaxwell.blogspot.com
allanfersmaxwell.blogspot.com
How to Calculate Delta H of Neutralization
Solved From The Following Enthalpy Of Reaction Data And ΔHf∘ | Chegg.com
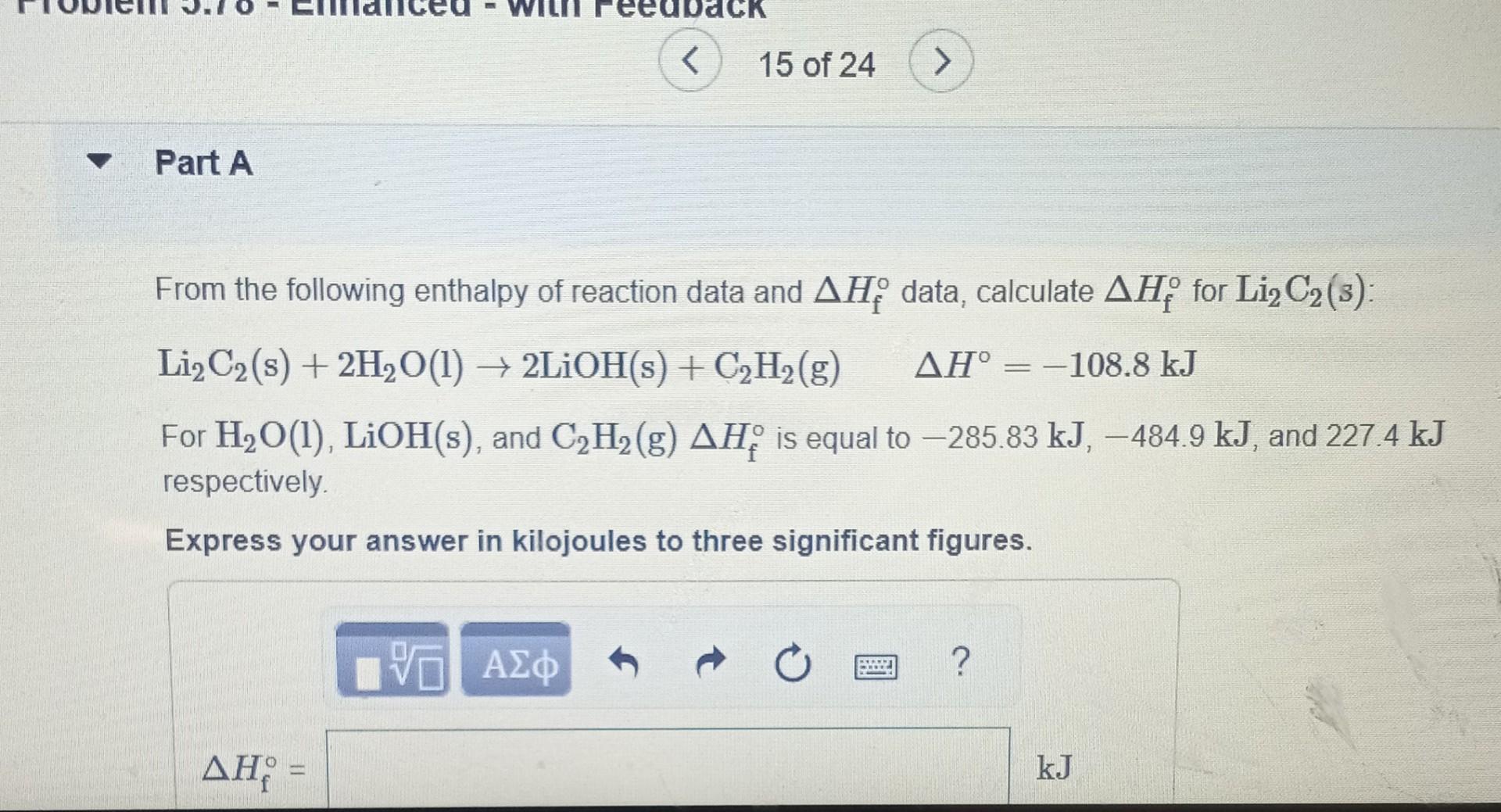 www.chegg.com
www.chegg.com
Solved From the following enthalpy of reaction data and ΔHf∘ | Chegg.com
How To Calculate Delta H Of Neutralization
 allanfersmaxwell.blogspot.com
allanfersmaxwell.blogspot.com
How to Calculate Delta H of Neutralization
Calculate The Enthalpy (ΔH∘) Of The Following | Chegg.com
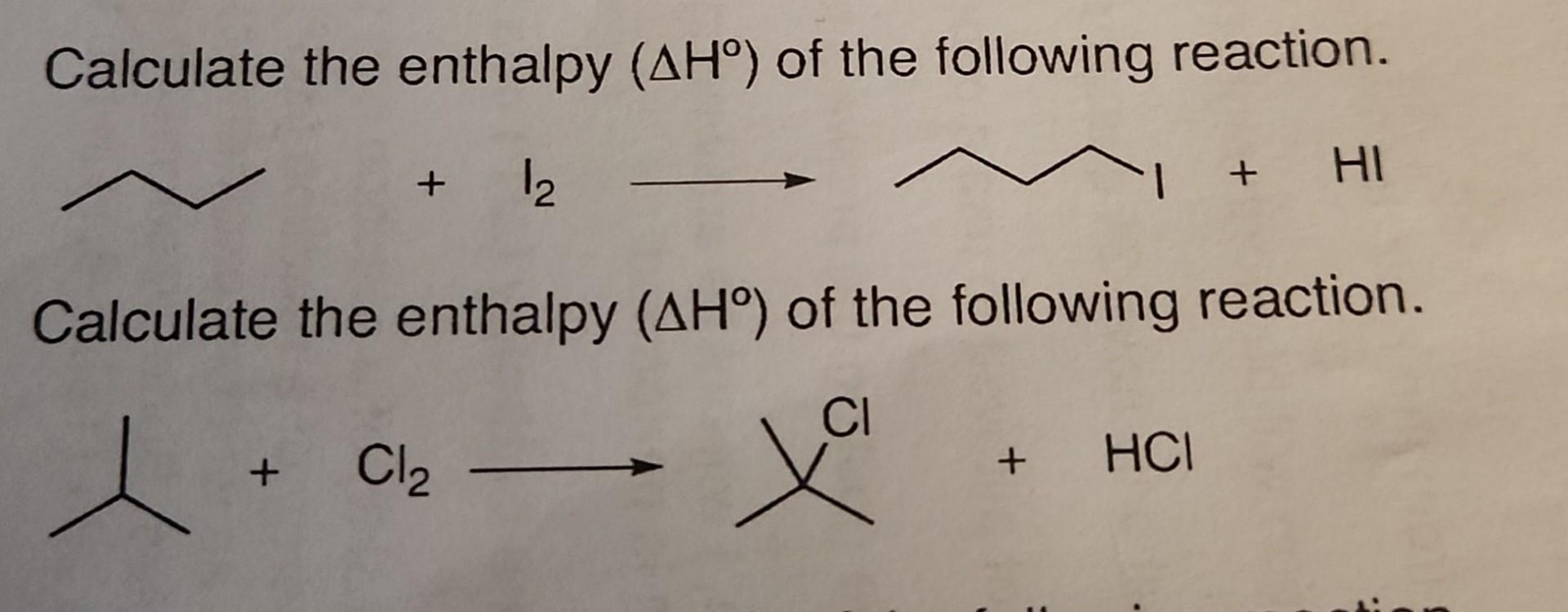 www.chegg.com
www.chegg.com
Calculate the enthalpy (ΔH∘) of the following | Chegg.com
Solved Calculate The Standard Enthalpy Of Reaction, ΔH°, at | Chegg.com
 www.chegg.com
www.chegg.com
Solved Calculate the standard enthalpy of reaction, ΔH°, at | Chegg.com
How do you calculate delta h in chemistry?. How do you calculate delta h in chemistry?. Solved part a: calculate delta h for the reaction between
