Galvanic corrosion is a type of corrosion that occurs when two dissimilar metals are in contact with each other in the presence of an electrolyte. This can lead to the degradation of one of the metals, resulting in significant damage and potentially costly repairs. To understand and prevent galvanic corrosion, it's essential to refer to a galvanic corrosion chart, also known as a galvanic series chart. This chart ranks metals based on their nobility, with the most noble metals being the least reactive and the least noble metals being the most reactive. In this article, we'll break down the key aspects of a galvanic corrosion chart and how it can be applied in real-world situations.
1. Understanding the Galvanic Series
The galvanic series is a ranking of metals based on their nobility, with the most noble metals at the top and the least noble metals at the bottom. The series is typically arranged in order of decreasing nobility, with metals such as gold and platinum being the most noble and metals such as magnesium and zinc being the least noble. This ranking is crucial in determining the likelihood of galvanic corrosion, as the greater the difference in nobility between two metals, the greater the risk of corrosion.
2. Identifying Compatible Metals
One of the primary uses of a galvanic corrosion chart is to identify compatible metals that can be used together in a given application. By selecting metals that are close together in the galvanic series, the risk of galvanic corrosion can be minimized. For example, if you need to use a fastener to join two metal components, selecting a fastener made from a metal that is similar in nobility to the components can help prevent corrosion.
3. Understanding the Role of the Electrolyte
An electrolyte is a substance that facilitates the flow of electrical current between two metals. In the context of galvanic corrosion, the electrolyte is typically a liquid, such as seawater or moisture, that comes into contact with the metals. The presence of an electrolyte is essential for galvanic corrosion to occur, as it allows the transfer of ions between the metals. Understanding the role of the electrolyte is critical in preventing galvanic corrosion, as it can help identify situations where corrosion is likely to occur.
4. Recognizing the Importance of Metal Thickness
The thickness of the metals involved can also play a significant role in determining the likelihood of galvanic corrosion. Thicker metals are less susceptible to corrosion than thinner metals, as they have a lower surface-to-volume ratio. This means that the anodic metal (the metal that is more prone to corrosion) will corrode more slowly, reducing the overall risk of damage.
5. Considering the Effects of Oxygen
Oxygen can also play a significant role in galvanic corrosion, particularly in situations where the metals are exposed to air. Oxygen can accelerate the corrosion process by reacting with the anodic metal, increasing the rate of corrosion. In applications where oxygen is present, it's essential to take steps to prevent or minimize its effects, such as using a coating or sealant to exclude oxygen.
6. Understanding the Difference Between Galvanic and Electrolytic Corrosion
While galvanic corrosion and electrolytic corrosion are both types of corrosion that occur in the presence of an electrolyte, they are distinct phenomena. Galvanic corrosion occurs when two dissimilar metals are in contact, while electrolytic corrosion occurs when an electric current is applied to a metal in the presence of an electrolyte. Understanding the difference between these two types of corrosion is essential in identifying and preventing corrosion in a given application.
7. Applying the Galvanic Series in Real-World Situations
The galvanic series can be applied in a wide range of real-world situations, from construction and engineering to manufacturing and maintenance. By understanding the nobility of different metals and how they interact with each other, professionals can design and build systems that minimize the risk of galvanic corrosion. For example, in the construction of a bridge, the galvanic series can be used to select compatible metals for the bridge's structural components, reducing the risk of corrosion and extending the bridge's lifespan.
8. Using Coatings and Sealants to Prevent Corrosion
Coatings and sealants can be used to prevent or minimize galvanic corrosion by excluding oxygen and moisture from the metals. These coatings can be applied to the metals themselves or to the surrounding environment, depending on the specific application. By preventing the flow of electrical current between the metals, coatings and sealants can reduce the risk of corrosion and protect the metals from damage.
9. Monitoring and Maintaining Systems to Prevent Corrosion
Regular monitoring and maintenance are critical in preventing galvanic corrosion. By inspecting systems and components regularly, professionals can identify potential corrosion risks and take steps to address them before they become major problems. This can include cleaning and inspecting the metals, applying coatings or sealants, and replacing corroded components.
10. Continuous Learning and Improvement
Finally, it's essential to recognize that galvanic corrosion is a complex phenomenon that requires continuous learning and improvement. As new materials and technologies are developed, the risk of galvanic corrosion can change, and professionals must stay up-to-date with the latest research and best practices to ensure that they are taking the necessary steps to prevent corrosion. By combining knowledge of the galvanic series with practical experience and ongoing education, professionals can minimize the risk of galvanic corrosion and ensure the longevity and reliability of the systems and components they design and build.
If you are searching about Printable Galvanic Corrosion Chart - Free Printable Charts you've visit to the right web. We have 10 Pics about Printable Galvanic Corrosion Chart - Free Printable Charts like Corrosion Guide Galvanic Chart Summary | PDF | Corrosion | Metals, Galvanic Corrosion [with Chart] - EngineerExcel and also Corrosion Guide Galvanic Chart Summary | PDF | Corrosion | Metals. Here you go:
Printable Galvanic Corrosion Chart - Free Printable Charts
 gea.edu.ge
gea.edu.ge
Printable Galvanic Corrosion Chart - Free Printable Charts
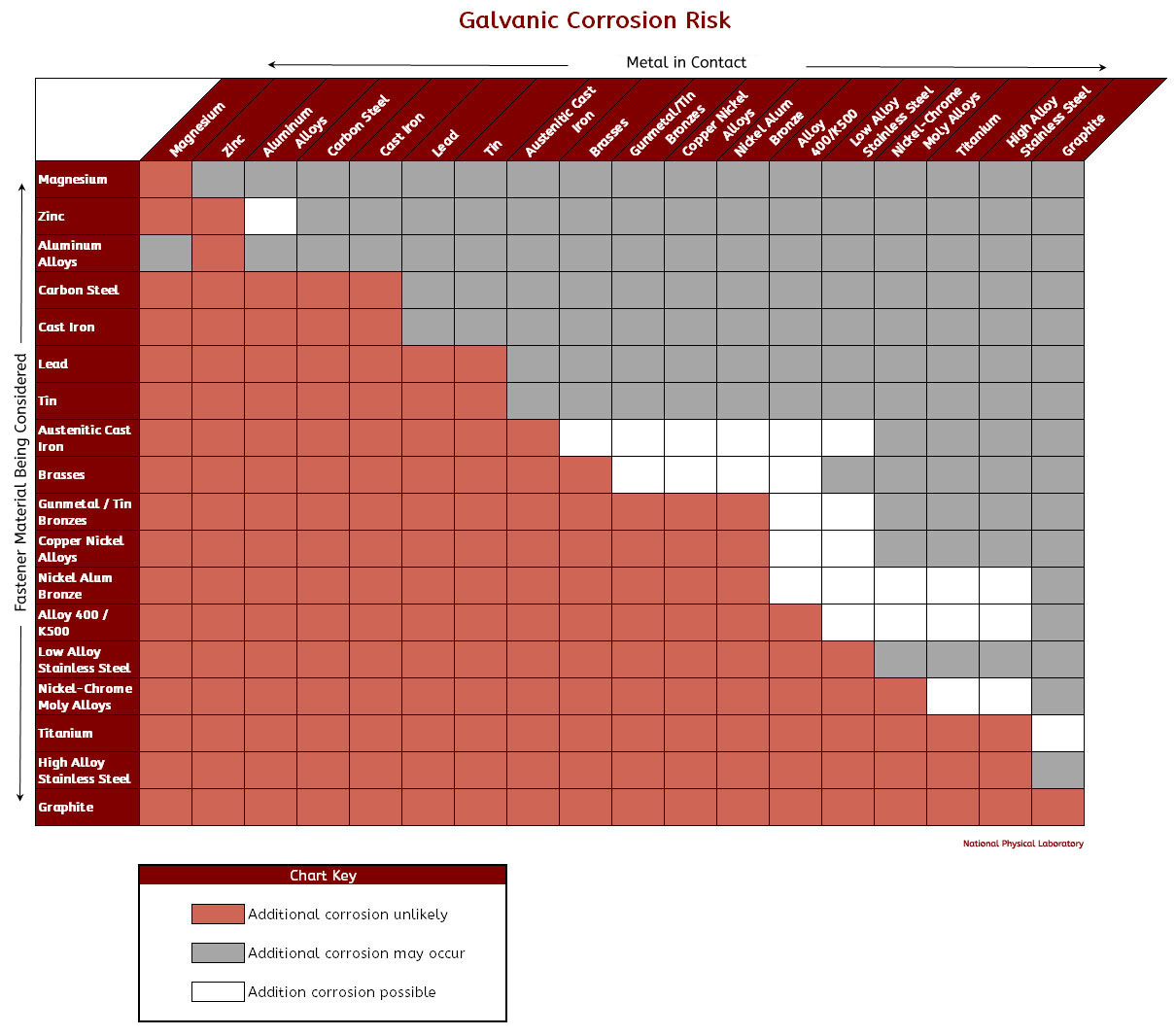
Galvanic Corrosion Chart
 www.extreme-bolt.com
www.extreme-bolt.com
Galvanic Corrosion Chart
Galvanic Corrosion Chart | Corrosion, Chart, Compatibility Chart
 www.pinterest.com
www.pinterest.com
Galvanic Corrosion Chart | Corrosion, Chart, Compatibility chart
Galvanic Corrosion And Red Plague | Lectromec
 lectromec.com
lectromec.com
Galvanic Corrosion and Red Plague | Lectromec
Corrosion Guide Galvanic Chart Summary | PDF | Corrosion | Metals
 www.scribd.com
www.scribd.com
Corrosion Guide Galvanic Chart Summary | PDF | Corrosion | Metals
Galvanic Corrosion: Causes, Effects, And Prevention Methods - Leading
 ssmalloys.com
ssmalloys.com
Galvanic Corrosion: Causes, Effects, and Prevention Methods - Leading ...
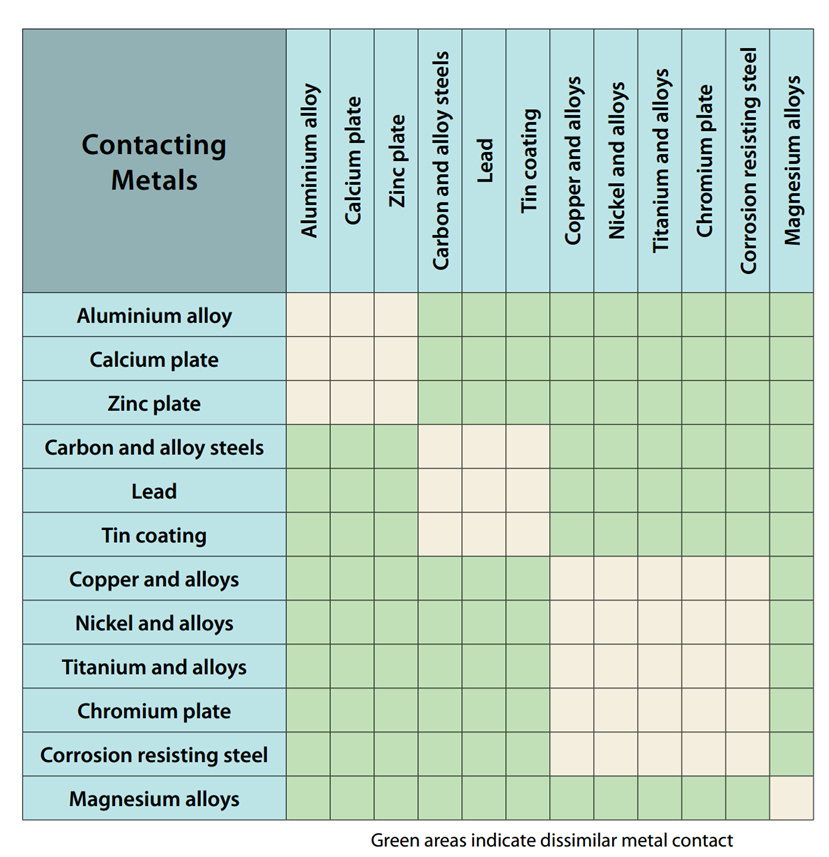
Galvanic Corrosion [with Chart] - EngineerExcel
![Galvanic Corrosion [with Chart] - EngineerExcel](https://engineerexcel.com/wp-content/uploads/2023/03/galvanic-corrosion-chart.webp) engineerexcel.com
engineerexcel.com
Galvanic Corrosion [with Chart] - EngineerExcel
Galvanic Corrosion [with Chart] - EngineerExcel
![Galvanic Corrosion [with Chart] - EngineerExcel](https://engineerexcel.com/wp-content/uploads/2023/03/galvanic-compatibility.webp) engineerexcel.com
engineerexcel.com
Galvanic Corrosion [with Chart] - EngineerExcel
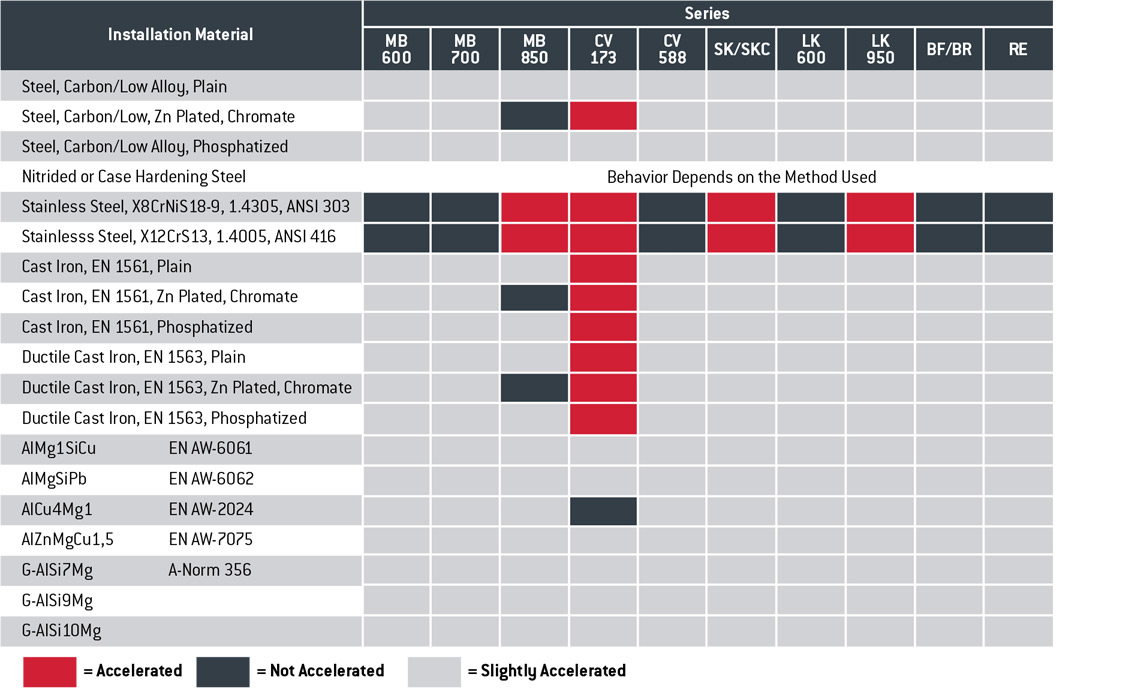
Galvanic Corrosion Chart And Guidelines For The Selection Of Fasteners
 www.fastenersystems.com
www.fastenersystems.com
Galvanic Corrosion Chart and Guidelines for the Selection of Fasteners ...
Printable Galvanic Corrosion Chart - Free Printable Charts
 gea.edu.ge
gea.edu.ge
Printable Galvanic Corrosion Chart - Free Printable Charts
Galvanic corrosion chart. galvanic corrosion: causes, effects, and prevention methods. Galvanic corrosion chart and guidelines for the selection of fasteners
