Dilutions are a fundamental concept in various fields, including chemistry, biology, and pharmacology. Calculating dilutions accurately is crucial to ensure the desired concentration of a substance in a solution. In this article, we will explore the steps to calculate dilutions and provide a comprehensive guide on the subject. From understanding the basics of dilutions to calculating serial dilutions, we will cover it all.
1. Understanding the Basics of Dilutions
Dilutions involve the reduction of the concentration of a substance in a solution by adding a solvent. The initial concentration of the substance is called the stock concentration, and the final concentration after dilution is called the working concentration. To calculate dilutions, you need to know the initial volume and concentration of the stock solution and the desired final concentration and volume of the working solution.
2. Calculating Dilutions Using the Formula
The formula to calculate dilutions is M1V1 = M2V2, where M1 is the initial concentration, V1 is the initial volume, M2 is the final concentration, and V2 is the final volume. By rearranging this formula, you can solve for the final volume or concentration. For example, if you want to calculate the final volume of a solution with a desired concentration, you can use the formula V2 = M1V1 / M2.
3. Calculating Serial Dilutions
Serial dilutions involve a series of dilutions, where each dilution is made from the previous one. To calculate serial dilutions, you need to know the initial concentration and volume of the stock solution and the desired final concentration and volume of the working solution. You can use the formula M1V1 = M2V2 for each dilution step, and then repeat the process for each subsequent dilution.
4. Understanding Concentration Units
Concentration units are crucial in calculating dilutions. The most common concentration units are molarity (M), molality (m), and percentage (%). Molarity is defined as the number of moles of solute per liter of solution, while molality is defined as the number of moles of solute per kilogram of solvent. Percentage is defined as the mass of solute per 100 grams of solution.
5. Calculating Dilutions with Percentage Concentration
When working with percentage concentrations, you need to convert the percentage to a decimal by dividing by 100. For example, 10% is equivalent to 0.1. You can then use the formula M1V1 = M2V2 to calculate the dilution. For example, if you want to calculate the final volume of a solution with a desired concentration of 5% from a stock solution of 10%, you can use the formula V2 = M1V1 / M2.
6. Calculating Dilutions with Molarity
When working with molar concentrations, you need to know the molar mass of the solute to calculate the number of moles. You can then use the formula M1V1 = M2V2 to calculate the dilution. For example, if you want to calculate the final volume of a solution with a desired concentration of 0.5 M from a stock solution of 1 M, you can use the formula V2 = M1V1 / M2.
7. Calculating Dilutions with Molality
When working with molal concentrations, you need to know the mass of the solvent to calculate the number of moles. You can then use the formula M1V1 = M2V2 to calculate the dilution. For example, if you want to calculate the final volume of a solution with a desired concentration of 0.5 m from a stock solution of 1 m, you can use the formula V2 = M1V1 / M2.
8. Using Dilution Factors to Calculate Dilutions
Dilution factors are a convenient way to calculate dilutions. A dilution factor is the ratio of the initial concentration to the final concentration. For example, a dilution factor of 10 means that the final concentration is 1/10 of the initial concentration. You can use dilution factors to calculate the final volume or concentration of a solution.
9. Calculating Dilutions with Multiple Dilution Steps
In some cases, you may need to perform multiple dilution steps to achieve the desired concentration. To calculate the final concentration, you need to multiply the dilution factors for each step. For example, if you perform a 1:10 dilution followed by a 1:5 dilution, the overall dilution factor is 1:50.
10. Verifying Dilution Calculations
It's essential to verify your dilution calculations to ensure accuracy. You can do this by checking your calculations with a calculator or by using online dilution calculators. Additionally, you can use a laboratory balance to measure the mass of the solute and solvent to ensure that your calculations are correct.
If you are looking for What is dilutions? How to calculate it? - Dilutions KABrown 6/ It is you've visit to the right place. We have 10 Images about What is dilutions? How to calculate it? - Dilutions KABrown 6/ It is like Dilutions Demystified: A Guide to Common Dilution Methods and, How To Calculate Dilutions Microbiology at Alma Kathryn blog and also How To Write Dilutions at Judy Moody blog. Here you go:
What Is Dilutions? How To Calculate It? - Dilutions KABrown 6/ It Is
 www.studocu.com
www.studocu.com
What is dilutions? How to calculate it? - Dilutions KABrown 6/ It is ...
How To Calculate Dilutions Microbiology At Alma Kathryn Blog
 storage.googleapis.com
storage.googleapis.com
How To Calculate Dilutions Microbiology at Alma Kathryn blog
Dilutions Demystified: A Guide To Common Dilution Methods And
 www.scribd.com
www.scribd.com
Dilutions Demystified: A Guide to Common Dilution Methods and ...
How To Write Dilutions At Judy Moody Blog
How To Write Dilutions at Judy Moody blog
How To Calculate Dilutions Microbiology At Aaron Battye Blog
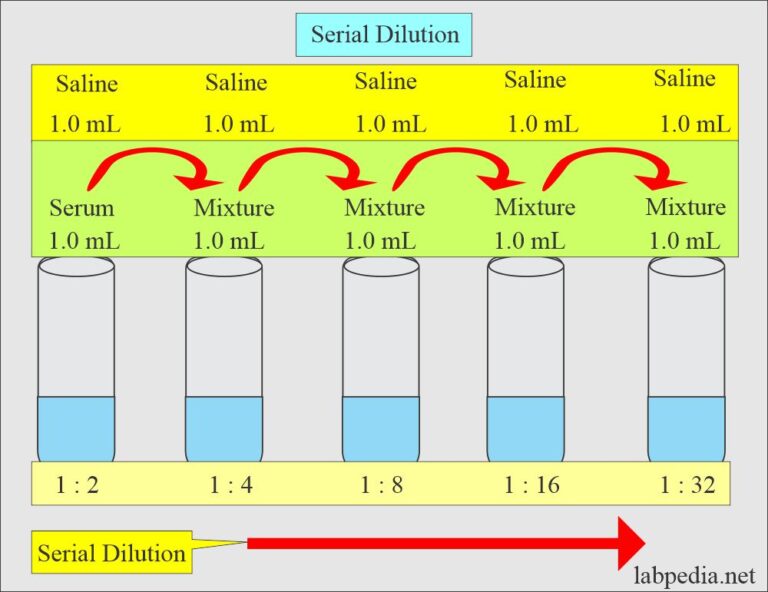 storage.googleapis.com
storage.googleapis.com
How To Calculate Dilutions Microbiology at Aaron Battye blog
Concentrations And Dilutions | MME
 mmerevise.co.uk
mmerevise.co.uk
Concentrations and Dilutions | MME
How Do You Calculate Dilution Factor?
 scienceoxygen.com
scienceoxygen.com
How do you calculate dilution factor?
How To Calculate Dilutions Microbiology At Alma Kathryn Blog
 storage.googleapis.com
storage.googleapis.com
How To Calculate Dilutions Microbiology at Alma Kathryn blog
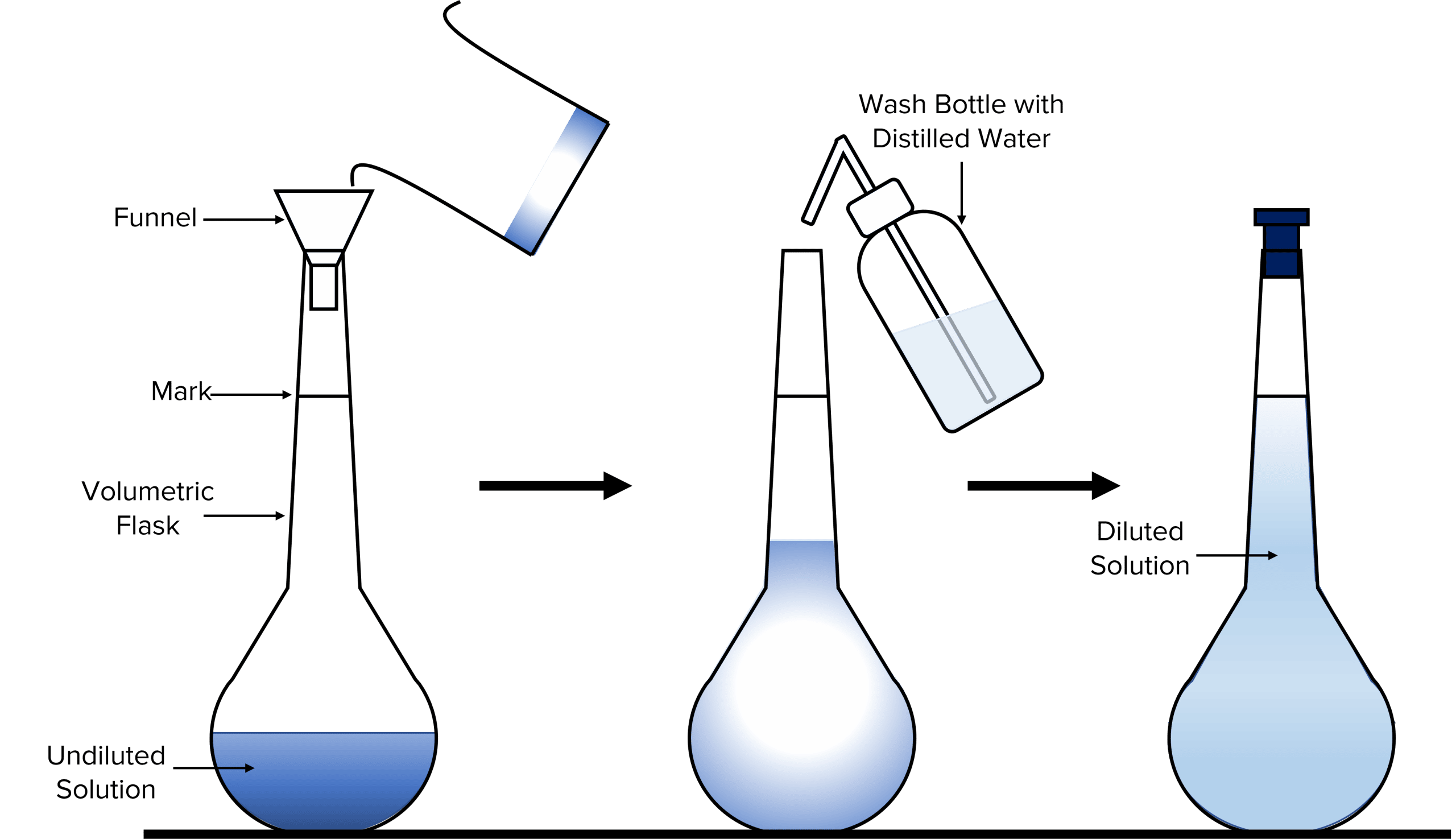
How To Make Dilutions In Lab At Drew Ballard Blog
 dxobyjbye.blob.core.windows.net
dxobyjbye.blob.core.windows.net
How To Make Dilutions In Lab at Drew Ballard blog
Dilutions Used In The Real World At Patrick Drago Blog
 storage.googleapis.com
storage.googleapis.com
Dilutions Used In The Real World at Patrick Drago blog
How to make dilutions in lab at drew ballard blog. How to write dilutions at judy moody blog. What is dilutions? how to calculate it?
