When it comes to chemistry, one of the most important concepts to grasp is the oxidation number, also known as the oxidation state. It's a crucial aspect of understanding how elements interact with each other and form compounds. In this post, we'll delve into the world of calculator oxidation numbers and explore how they can help you master chemistry. Whether you're a student or just a curious learner, this list will guide you through the essentials of oxidation numbers and how to calculate them.
1. Understanding Oxidation Numbers
Oxidation numbers are a way to keep track of the number of electrons an atom has gained or lost when it forms a compound. This number can be positive, negative, or zero, depending on the element and its position in the periodic table. To calculate the oxidation number, you need to know the rules, which we'll explore in the following points.
2. Rules for Assigning Oxidation Numbers
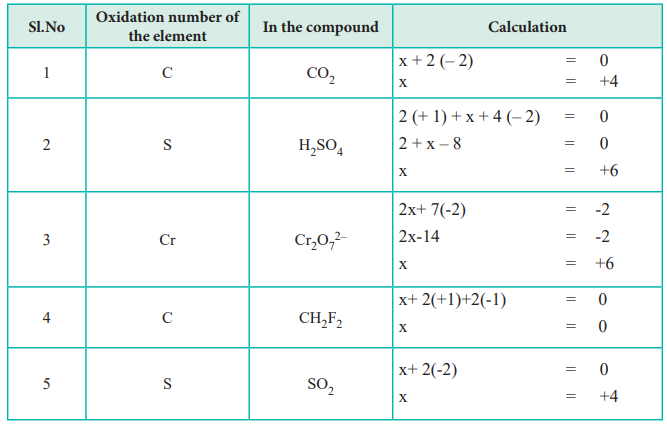
The first rule is that the oxidation number of a free element is always zero. This means that when an element is not combined with any other element, its oxidation number is zero. The second rule is that the oxidation number of a monatomic ion is equal to its charge. For example, the oxidation number of sodium (Na) in the compound NaCl is +1, because it has lost one electron to become a positive ion.
3. Calculating Oxidation Numbers in Compounds
In compounds, the oxidation number of each element can be calculated by considering the other elements present. For example, in the compound H2O, the oxidation number of hydrogen (H) is +1, and the oxidation number of oxygen (O) is -2. This is because oxygen is more electronegative than hydrogen, so it pulls the electrons towards itself, resulting in a negative oxidation number.
4. Oxidation Numbers of Transition Metals
Transition metals can have multiple oxidation numbers, depending on the compound they're in. For example, iron (Fe) can have an oxidation number of +2 or +3, depending on the ligands attached to it. This is because transition metals have multiple valence electrons, which can be lost or gained to form different compounds.
5. Calculating Oxidation Numbers in Complex Compounds
In complex compounds, the oxidation number of each element can be calculated by considering the ligands and the central metal atom. For example, in the compound [Fe(CN)6]3-, the oxidation number of iron is +3, because it has lost three electrons to form a stable compound with the cyanide ligands.
6. Using a Calculator to Determine Oxidation Numbers
While it's possible to calculate oxidation numbers by hand, using a calculator can make the process much faster and more accurate. There are many online calculators available that can help you determine the oxidation number of an element in a compound. These calculators use algorithms to apply the rules of oxidation numbers and provide the correct answer.
7. Importance of Oxidation Numbers in Chemistry
Oxidation numbers are crucial in chemistry because they help us understand the properties and behavior of elements and compounds. They can be used to predict the reactivity of a compound, its stability, and its potential to undergo certain reactions. By mastering oxidation numbers, you can gain a deeper understanding of chemistry and develop a strong foundation for further study.
8. Common Mistakes to Avoid When Calculating Oxidation Numbers
One common mistake to avoid when calculating oxidation numbers is to forget to consider the charge of the compound. This can lead to incorrect calculations and a lack of understanding of the underlying chemistry. Another mistake is to assume that the oxidation number of an element is always the same, when in fact it can vary depending on the compound and the other elements present.
9. Practice Exercises to Master Oxidation Numbers
To become proficient in calculating oxidation numbers, it's essential to practice with exercises and examples. You can find many online resources that provide practice problems and quizzes to help you test your understanding of oxidation numbers. By working through these exercises, you can develop your skills and build confidence in your ability to calculate oxidation numbers.
10. Conclusion and Final Tips
In conclusion, oxidation numbers are a fundamental concept in chemistry that can seem intimidating at first, but with practice and patience, you can master them. Remember to always follow the rules, consider the charge of the compound, and practice with exercises to become proficient. With these tips and a solid understanding of oxidation numbers, you'll be well on your way to exceling in chemistry and unlocking the secrets of the periodic table.
If you are searching about Oxidation Number Calculator - Calculator Academy you've came to the right page. We have 10 Pictures about Oxidation Number Calculator - Calculator Academy like Oxidation Number Calculator - Calculator Academy, GitHub - totwjfakd/oxidationNumberCalculator and also Oxidation Number Calculator - Calculator Academy. Here it is:
Oxidation Number Calculator - Calculator Academy
 calculator.academy
calculator.academy
Oxidation Number Calculator - Calculator Academy
Oxidation Number Calculator
 fity.club
fity.club
Oxidation Number Calculator
Oxidation Number Calculator - Yes! Calculator
 www.yescalculator.com
www.yescalculator.com
Oxidation Number Calculator - Yes! Calculator
Oxidation Number Calculator
 fity.club
fity.club
Oxidation Number Calculator
Oxidation Number Calculator
 fity.club
fity.club
Oxidation Number Calculator
Oxidation Number Calculator
 fity.club
fity.club
Oxidation Number Calculator
GitHub - Riadan710/Oxidation-Number-Calculator: 2019년 1학기 화학 세특용 화학 산화수
GitHub - riadan710/Oxidation-Number-Calculator: 2019년 1학기 화학 세특용 화학 산화수 ...
GitHub - Totwjfakd/oxidationNumberCalculator
GitHub - totwjfakd/oxidationNumberCalculator
Oxidation Number Calculator
 fity.club
fity.club
Oxidation Number Calculator
Oxidation Numbers | Master Electron Transfer - Get Started Now — CHEMDUNN
 www.chemdunn.com
www.chemdunn.com
Oxidation Numbers | Master Electron Transfer - Get Started Now — CHEMDUNN
oxidation number calculator. Oxidation numbers. Oxidation number calculator
