When it comes to chemistry, understanding the concentration of a solution is crucial. One way to express concentration is through molality, which is defined as the number of moles of solute per kilogram of solvent. Calculating molality can seem intimidating, but it's actually quite straightforward once you know the steps. In this post, we'll break down the process into manageable parts, so you can confidently calculate molality like a pro.
1. Define Molality and Its Importance
Molality is a measure of the concentration of a solution, and it's essential in various chemical calculations, such as determining the freezing point depression or boiling point elevation of a solution. It's also used to calculate the vapor pressure of a solution. Understanding molality is vital in chemistry, and it's used in various fields, including physics, biology, and engineering.
2. Identify the Given Values
To calculate molality, you need to know the number of moles of solute and the mass of the solvent in kilograms. You can find these values in the problem statement or by measuring them experimentally. Make sure to note the units of each value, as they will be crucial in the calculation.
3. Calculate the Number of Moles of Solute
The number of moles of solute can be calculated using the formula: moles = mass of solute / molar mass of solute. You can find the molar mass of the solute on the periodic table or in a reference book. Be sure to use the correct units, as the calculation requires the mass of the solute in grams.
4. Determine the Mass of the Solvent
The mass of the solvent is typically given in the problem statement or can be measured experimentally. Make sure to note the units of the mass, as it should be in kilograms. If the mass is given in grams, you'll need to convert it to kilograms by dividing by 1000.
5. Calculate the Molality
Now that you have the number of moles of solute and the mass of the solvent, you can calculate the molality using the formula: molality = moles of solute / mass of solvent (in kg). This will give you the concentration of the solution in moles of solute per kilogram of solvent.
6. Consider the Units
When calculating molality, it's essential to consider the units of each value. The number of moles of solute should be in moles (mol), and the mass of the solvent should be in kilograms (kg). The resulting molality will be in moles of solute per kilogram of solvent (m).
7. Check for Significant Figures
When calculating molality, it's crucial to consider significant figures. The number of significant figures in the answer should be the same as the least number of significant figures in the given values. This ensures that the answer is accurate and reliable.
8. Apply Molality in Real-World Scenarios
Molality is not just a theoretical concept; it has numerous real-world applications. For example, it's used in the production of antifreeze, where the molality of the solution determines its freezing point. It's also used in the manufacture of foods, such as jams and jellies, where the molality of the sugar solution affects the final product's texture and consistency.
9. Practice with Example Problems
To become proficient in calculating molality, practice with example problems. Start with simple problems and gradually move on to more complex ones. This will help you develop a deeper understanding of the concept and build your confidence in calculating molality.
10. Review and Refine Your Understanding
Finally, review and refine your understanding of molality by revisiting the concepts and formulas regularly. This will help reinforce your knowledge and ensure that you can apply it accurately in various situations. With practice and review, you'll become proficient in calculating molality and be able to tackle complex chemical problems with ease.
If you are looking for What is Molality? Definition & Simple Calculations - Biology Reader you've visit to the right web. We have 10 Pics about What is Molality? Definition & Simple Calculations - Biology Reader like Molality: Formula, Calculation, Advantages, Limitations, Molality — Definition & Calculation - Expii and also Molality | PDF. Here it is:
What Is Molality? Definition & Simple Calculations - Biology Reader
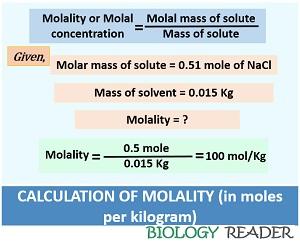 biologyreader.com
biologyreader.com
What is Molality? Definition & Simple Calculations - Biology Reader
Molality | PDF
 www.scribd.com
www.scribd.com
Molality | PDF
Molality Calculator - Inch Calculator
 www.inchcalculator.com
www.inchcalculator.com
Molality Calculator - Inch Calculator
Molality — Definition & Calculation - Expii
 www.expii.com
www.expii.com
Molality — Definition & Calculation - Expii
Molality Calculator | Definition | Formula
 www.omnicalculator.com
www.omnicalculator.com
Molality Calculator | Definition | Formula
Molality: Formula, Calculation, Advantages, Limitations
 scienceinfo.com
scienceinfo.com
Molality: Formula, Calculation, Advantages, Limitations
Molality - Chemistry Steps
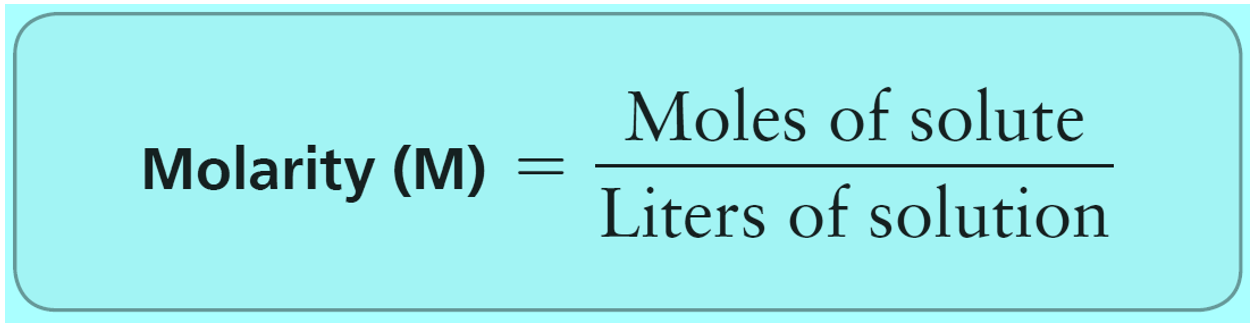 general.chemistrysteps.com
general.chemistrysteps.com
Molality - Chemistry Steps
What Is Molality? Definition & Simple Calculations - Biology Reader
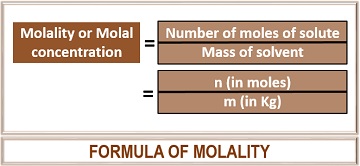 biologyreader.com
biologyreader.com
What is Molality? Definition & Simple Calculations - Biology Reader
Molality Calculator - Sum SQ
 sumsq.com
sumsq.com
Molality Calculator - Sum SQ
Molality Formula: Learn Some Quick Calculation Tips
 entechonline.com
entechonline.com
Molality Formula: Learn some Quick Calculation Tips
molality: formula, calculation, advantages, limitations. molality calculator. Molality formula: learn some quick calculation tips
