When it comes to chemistry, one of the most crucial concepts to grasp is the idea of a limiting reagent. This is the substance that will be completely consumed in a reaction, thereby determining the amount of product formed. In Asian cultures, attention to detail and respect for tradition are highly valued, and calculating the limiting reagent is no exception. Here are the steps to follow when determining the limiting reagent in a chemical reaction, presented with the clarity and precision that Asian cultures are known for.
1. Write Down the Balanced Chemical Equation
The first step in calculating the limiting reagent is to write down the balanced chemical equation for the reaction. This equation should include the reactants, products, and the coefficients (numbers in front of the formulas of reactants or products) that balance the equation. For example, the equation for the combustion of methane (CH4) is CH4 + 2O2 → CO2 + 2H2O. The coefficients here are 1 for CH4, 2 for O2, 1 for CO2, and 2 for H2O.
2. Identify the Given Information
Next, identify the given information about the reactants, such as their masses or volumes. This information will be used to calculate the number of moles of each reactant. For instance, if you have 10 grams of CH4 and 20 grams of O2, you will need to calculate the number of moles of each using their molar masses (16.04 g/mol for CH4 and 32.00 g/mol for O2).
3. Calculate the Number of Moles of Each Reactant
To calculate the number of moles of each reactant, divide the mass of the reactant by its molar mass. Using the example from step 2, for CH4: moles = mass / molar mass = 10 g / 16.04 g/mol = 0.623 mol. For O2: moles = mass / molar mass = 20 g / 32.00 g/mol = 0.625 mol.
4. Determine the Mole Ratio of the Reactants
From the balanced chemical equation, determine the mole ratio of the reactants. In the case of the combustion of methane, the mole ratio of CH4 to O2 is 1:2. This means for every mole of CH4, 2 moles of O2 are required for the reaction to proceed completely.
5. Compare the Mole Ratio of the Reactants
Compare the mole ratio from the balanced equation with the mole ratio of the reactants you have. For the combustion of methane with the moles calculated in step 3, you have 0.623 mol of CH4 and 0.625 mol of O2. The balanced equation requires 2 moles of O2 for every 1 mole of CH4, so the required moles of O2 for 0.623 mol of CH4 would be 0.623 * 2 = 1.246 mol of O2.
6. Identify the Limiting Reagent
The reactant that has fewer moles than required for the reaction to proceed completely according to the balanced equation is the limiting reagent. Since you only have 0.625 mol of O2, which is less than the 1.246 mol required for 0.623 mol of CH4, O2 is the limiting reagent in this scenario.
7. Calculate the Maximum Amount of Product Formed
With the limiting reagent identified, you can calculate the maximum amount of product that can be formed. Using the stoichiometry of the reaction, the number of moles of the limiting reagent is used to find the moles of product. For the combustion of methane, since O2 is the limiting reagent and it produces CO2 in a 1:1 ratio with O2, the maximum moles of CO2 that can be produced is equal to the moles of O2, which is 0.625 mol.
8. Consider the Theoretical Yield
The theoretical yield of a reaction is the amount of product that would be obtained if the reaction were 100% efficient. This can be calculated by converting the moles of product (from step 7) into mass using the molar mass of the product. For CO2, with a molar mass of 44.01 g/mol, the theoretical yield would be 0.625 mol * 44.01 g/mol = 27.51 grams.
9. Account for Reaction Conditions
In real-world reactions, conditions such as temperature, pressure, and the presence of catalysts can affect the reaction rate and efficiency. These conditions should be optimized to ensure the reaction proceeds as close to 100% efficiency as possible, though in practice, the actual yield will often be less than the theoretical yield due to various losses and inefficiencies.
10. Practice and Apply
Like any skill, calculating the limiting reagent and determining the theoretical yield of a reaction becomes easier with practice. Applying these principles to various chemical reactions helps deepen understanding and improves the ability to solve complex problems in chemistry, reflecting the Asian value of diligent practice and mastery of skills.
If you are searching about Step 1: Finding the Limiting Reagent - Three Steps to Success you've visit to the right page. We have 10 Images about Step 1: Finding the Limiting Reagent - Three Steps to Success like Finding the Limiting Reagent - Limiting Reagents, Finding the Limiting Reagent - Limiting Reagents and also How to Use the Limiting Reagent - Three Steps to Success. Here it is:
Step 1: Finding The Limiting Reagent - Three Steps To Success
 limitingreagents9.weebly.com
limitingreagents9.weebly.com
Step 1: Finding the Limiting Reagent - Three Steps to Success
Step 1: Finding The Limiting Reagent - Three Steps To Success
 limitingreagents9.weebly.com
limitingreagents9.weebly.com
Step 1: Finding the Limiting Reagent - Three Steps to Success
Limiting Reactants And Limiting Reagents | ChemTalk
 chemistrytalk.org
chemistrytalk.org
Limiting Reactants and Limiting Reagents | ChemTalk
Finding The Limiting Reagent - Limiting Reagents
 trevortry.weebly.com
trevortry.weebly.com
Finding the Limiting Reagent - Limiting Reagents
Step 1: Finding The Limiting Reagent - Three Steps To Success
 limitingreagents9.weebly.com
limitingreagents9.weebly.com
Step 1: Finding the Limiting Reagent - Three Steps to Success
Finding The Limiting Reagent - Limiting Reagents
 trevortry.weebly.com
trevortry.weebly.com
Finding the Limiting Reagent - Limiting Reagents
Ways To Calculate Limiting Reagent Youtube - Vrogue.co
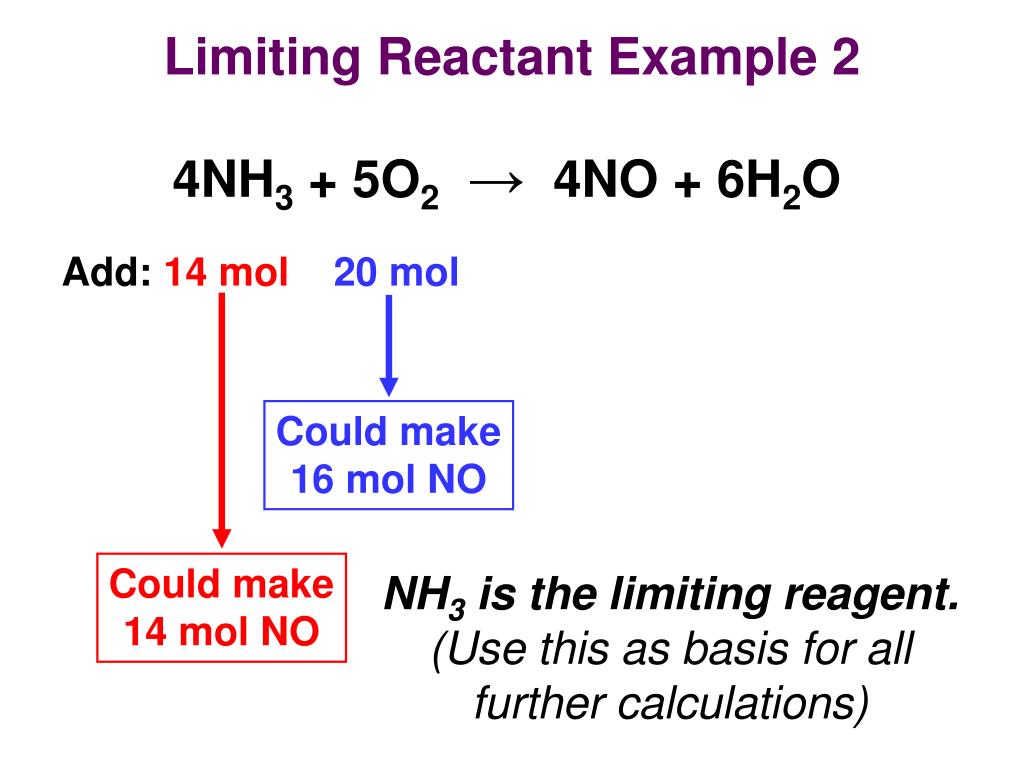 www.vrogue.co
www.vrogue.co
Ways To Calculate Limiting Reagent Youtube - vrogue.co
How To Use The Limiting Reagent - Three Steps To Success
 limitingreagents9.weebly.com
limitingreagents9.weebly.com
How to Use the Limiting Reagent - Three Steps to Success
How To Use The Limiting Reagent - Three Steps To Success
 limitingreagents9.weebly.com
limitingreagents9.weebly.com
How to Use the Limiting Reagent - Three Steps to Success
Finding The Limiting Reagent - Limiting Reagents
 trevortry.weebly.com
trevortry.weebly.com
Finding the Limiting Reagent - Limiting Reagents
Finding the limiting reagent. Step 1: finding the limiting reagent. How to use the limiting reagent
