Calculating the limiting reactant is a crucial step in determining the maximum amount of product that can be formed in a chemical reaction. This process involves identifying the reactant that will be completely consumed first, thereby limiting the amount of product that can be formed. In this article, we will explore the steps involved in calculating the limiting reactant, including the importance of stoichiometry, mole ratios, and reaction equations.
1. Understand the Concept of Limiting Reactant
The limiting reactant is the reactant that determines the maximum amount of product that can be formed in a chemical reaction. It is the reactant that will be completely consumed first, and its quantity will determine the amount of product that can be formed. Understanding this concept is essential in calculating the limiting reactant and determining the maximum amount of product that can be formed.
2. Write the Balanced Chemical Equation
To calculate the limiting reactant, it is essential to start with a balanced chemical equation. The balanced equation will provide the mole ratios of the reactants and products, which are critical in determining the limiting reactant. The equation should be balanced to ensure that the number of atoms of each element is the same on both the reactant and product sides.
3. Determine the Mole Ratios of the Reactants
Once the balanced chemical equation is written, the next step is to determine the mole ratios of the reactants. The mole ratios are calculated by dividing the coefficient of each reactant by the coefficient of the other reactant. These mole ratios are critical in determining the limiting reactant and will be used to compare the amounts of the reactants.
4. Calculate the Number of Moles of Each Reactant
To determine the limiting reactant, it is essential to calculate the number of moles of each reactant. The number of moles can be calculated using the formula: moles = mass / molar mass. This calculation will provide the amount of each reactant in moles, which will be used to compare the amounts of the reactants.
5. Compare the Mole Ratios of the Reactants
Once the number of moles of each reactant is calculated, the next step is to compare the mole ratios of the reactants. The mole ratios are compared to determine which reactant is the limiting reactant. The reactant with the smaller mole ratio is the limiting reactant, as it will be completely consumed first.
6. Determine the Limiting Reactant
After comparing the mole ratios of the reactants, the limiting reactant can be determined. The limiting reactant is the reactant that has the smaller mole ratio and will be completely consumed first. This reactant will determine the maximum amount of product that can be formed.
7. Calculate the Maximum Amount of Product
Once the limiting reactant is determined, the maximum amount of product that can be formed can be calculated. The calculation involves multiplying the number of moles of the limiting reactant by the mole ratio of the product to the limiting reactant. This calculation will provide the maximum amount of product that can be formed.
8. Consider the Stoichiometry of the Reaction
The stoichiometry of the reaction is critical in calculating the limiting reactant. The stoichiometry refers to the quantitative relationships between the reactants and products in a chemical reaction. Understanding the stoichiometry of the reaction is essential in determining the limiting reactant and the maximum amount of product that can be formed.
9. Use Conversion Factors to Simplify Calculations
Conversion factors can be used to simplify calculations when determining the limiting reactant. Conversion factors are used to convert between different units, such as moles to grams or liters to moles. Using conversion factors can simplify calculations and reduce errors.
10. Verify the Calculation
Finally, it is essential to verify the calculation to ensure that the correct limiting reactant has been determined. The verification involves checking the calculation steps and ensuring that the correct mole ratios and conversion factors have been used. Verifying the calculation will ensure that the correct limiting reactant has been determined and the maximum amount of product that can be formed has been calculated accurately.
If you are searching about Limiting Reactant in the Stoichiometry of Chemical Reactions you've visit to the right place. We have 10 Pics about Limiting Reactant in the Stoichiometry of Chemical Reactions like Limiting Reactant Explained: Step-by-Step Guide & Practice Problems, Limiting Reactant Explained: Step-by-Step Guide & Practice Problems and also How To Calculate Limiting Reactant Chemistry. Here it is:
Limiting Reactant In The Stoichiometry Of Chemical Reactions
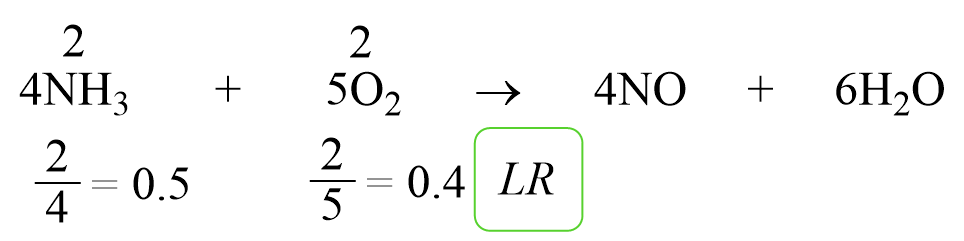 general.chemistrysteps.com
general.chemistrysteps.com
Limiting Reactant in the Stoichiometry of Chemical Reactions
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems
 www.chemdunn.com
www.chemdunn.com
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems ...
How To Calculate Limiting Reactant Chemistry Friday March 27
 fity.club
fity.club
How To Calculate Limiting Reactant Chemistry Friday March 27
How To Calculate Limiting Reactant Chemistry
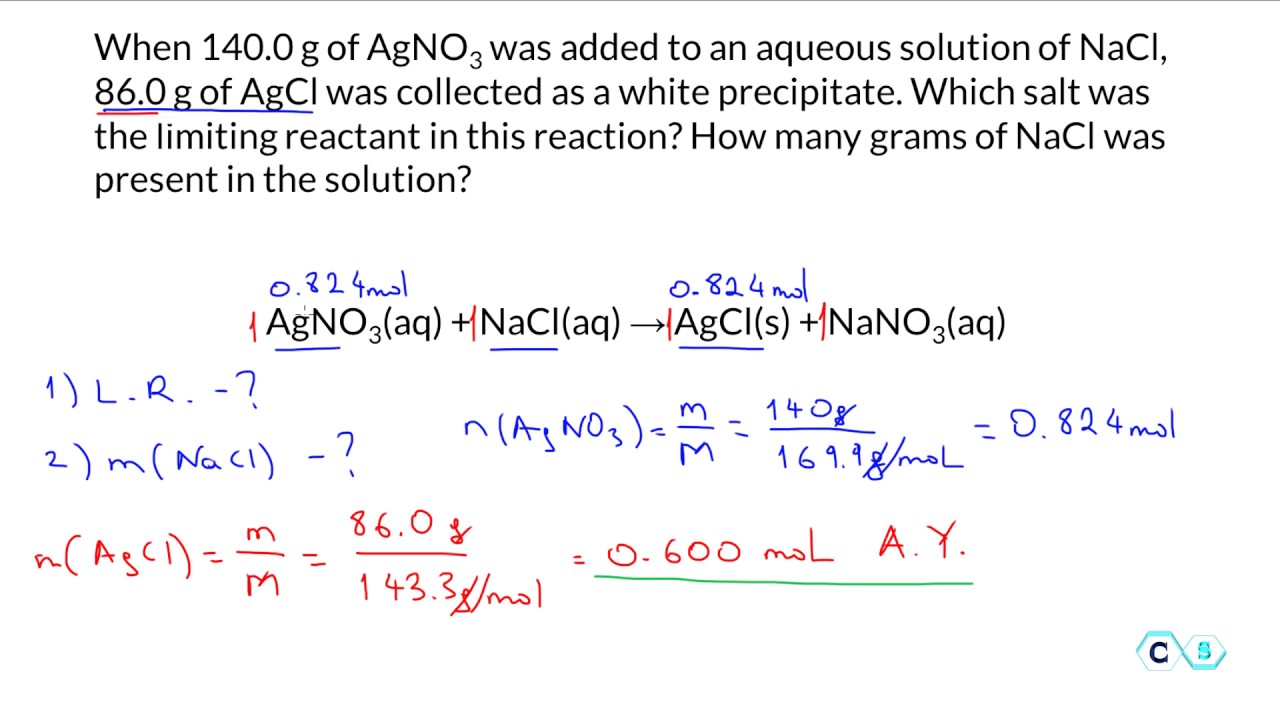 fity.club
fity.club
How To Calculate Limiting Reactant Chemistry
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems
 www.chemdunn.com
www.chemdunn.com
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems ...
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems
 www.chemdunn.com
www.chemdunn.com
Limiting Reactant Explained: Step-by-Step Guide & Practice Problems ...
Solved How Do You Calculate The Limiting Reactant? | Chegg.com
 www.chegg.com
www.chegg.com
Solved How do you calculate the limiting reactant? | Chegg.com
How To Calculate Limiting Reactant Chemistry
 fity.club
fity.club
How To Calculate Limiting Reactant Chemistry
Limiting Reactant | PDF
 www.scribd.com
www.scribd.com
Limiting Reactant | PDF
How To Calculate Limiting Reactant Chemistry
 fity.club
fity.club
How To Calculate Limiting Reactant Chemistry
How to calculate limiting reactant chemistry friday march 27. limiting reactant in the stoichiometry of chemical reactions. How to calculate limiting reactant chemistry
